Transitioning enantioselective indicator displacement assays for alpha-amino acids to protocols amenable to high-throughput screening
- PMID: 18714993
- PMCID: PMC7815441
- DOI: 10.1021/ja8038079
Transitioning enantioselective indicator displacement assays for alpha-amino acids to protocols amenable to high-throughput screening
Abstract
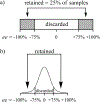
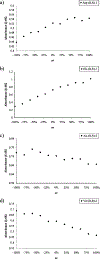
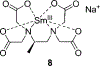
Enantioselective indicator displacement assays (eIDAs) for alpha-amino acids were conducted in a 96-well plate format to demonstrate the viability of the technique for the high-throughput screening (HTS) of enantiomeric excess (ee) values. Chiral receptors [Cu(II)(1)](2+) and [Cu(II)(2)](2+) with the indicator chrome azurol S were implemented for the eIDAs. Enantiomeric excess calibration curves were made using both receptors and then used to analyze true test samples. These results were compared to those previously obtained with a conventional UV-vis spectrophotometer, and they showed little to no loss of accuracy, while the speed of analysis was increased. A sample of valine of unknown ee was synthesized through an asymmetric reaction to produce a realistic reaction sample, which was analyzed using receptor [Cu(II)(1)](2+). The experimentally determined ee using our eIDA was compared to that obtained by chiral HPLC and (1)H NMR chiral shift reagent analysis. This gave errors of 4.7% and 12.0%, respectively. In addition to the use of ee calibration curves, an artificial neural network (ANN) was used to determine the % L-amino acid of the test samples and of the sample of valine of unknown ee from the asymmetric reaction. This method obtained errors of 5.9% and 2.2% compared to chiral HPLC and (1)H NMR chiral shift reagent analysis, respectively. The technique using calibration curves for the determination of ee on a 96-well plate allows one to determine 96 ee values in under a minute, enabling its use for HTS of asymmetric reactions with acceptable accuracy.
Figures




Similar articles
-
Using enantioselective indicator displacement assays to determine the enantiomeric excess of alpha-amino acids.J Am Chem Soc. 2008 Sep 17;130(37):12318-27. doi: 10.1021/ja803806c. Epub 2008 Aug 21. J Am Chem Soc. 2008. PMID: 18714996 Free PMC article.
-
Rapid optical methods for enantiomeric excess analysis: from enantioselective indicator displacement assays to exciton-coupled circular dichroism.Acc Chem Res. 2014 Jul 15;47(7):2212-21. doi: 10.1021/ar500147x. Epub 2014 Jun 3. Acc Chem Res. 2014. PMID: 24892802
-
High-throughput screening of identity, enantiomeric excess, and concentration using MLCT transitions in CD spectroscopy.J Am Chem Soc. 2008 Jul 23;130(29):9232-3. doi: 10.1021/ja803443j. Epub 2008 Jun 24. J Am Chem Soc. 2008. PMID: 18572934 Free PMC article.
-
Rapid determination of enantiomeric excess: a focus on optical approaches.Chem Soc Rev. 2012 Jan 7;41(1):448-79. doi: 10.1039/c1cs15135e. Epub 2011 Sep 5. Chem Soc Rev. 2012. PMID: 21892514 Review.
-
Enantioselective automultiplication of chiral molecules by asymmetric autocatalysis.Acc Chem Res. 2000 Jun;33(6):382-90. doi: 10.1021/ar9900820. Acc Chem Res. 2000. PMID: 10891056 Review.
Cited by
-
Using enantioselective indicator displacement assays to determine the enantiomeric excess of alpha-amino acids.J Am Chem Soc. 2008 Sep 17;130(37):12318-27. doi: 10.1021/ja803806c. Epub 2008 Aug 21. J Am Chem Soc. 2008. PMID: 18714996 Free PMC article.
-
Miniature high-throughput chemosensing of yield, ee, and absolute configuration from crude reaction mixtures.Sci Adv. 2016 Feb 12;2(2):e1501162. doi: 10.1126/sciadv.1501162. eCollection 2016 Feb. Sci Adv. 2016. PMID: 26933684 Free PMC article.
-
Chirality imprinting and direct asymmetric reaction screening using a stereodynamic Brønsted/Lewis acid receptor.Nat Commun. 2016 Aug 23;7:12539. doi: 10.1038/ncomms12539. Nat Commun. 2016. PMID: 27549926 Free PMC article.
-
Well Plate Circular Dichroism Reader for the Rapid Determination of Enantiomeric Excess.Chem Sci. 2014 Nov 1;5(11):4278-4282. doi: 10.1039/C4SC01641F. Chem Sci. 2014. PMID: 25386332 Free PMC article.
-
A fluorogenic aldehyde bearing a 1,2,3-triazole moiety for monitoring the progress of aldol reactions.J Org Chem. 2009 Mar 20;74(6):2417-24. doi: 10.1021/jo900013w. J Org Chem. 2009. PMID: 19222210 Free PMC article.
References
-
- Reetz MT Angew. Chem., Int. Ed 2001, 40, 284–310. - PubMed
-
- Tsukamoto M; Kagan HB Adv. Synth. Catal 2002, 344, 453–463.
-
- Traverse JF; Snapper ML Drug Discovery Today 2002, 7, 1002–1012. - PubMed
-
- Stambuli JP; Hartwig JF Curr. Opin. Chem. Biol 2003, 7, 420–426. - PubMed
-
- Collins AN; Sheldrake GNCJ Chirality in Industry; Wiley: New York/Chichester England, 1992.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

