Flavonoids redirect PIN-mediated polar auxin fluxes during root gravitropic responses
- PMID: 18718912
- PMCID: PMC2662185
- DOI: 10.1074/jbc.M710122200
Flavonoids redirect PIN-mediated polar auxin fluxes during root gravitropic responses
Abstract
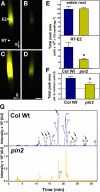
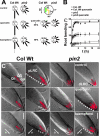
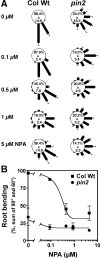
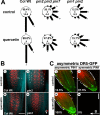
The rate, polarity, and symmetry of the flow of the plant hormone auxin are determined by the polar cellular localization of PIN-FORMED (PIN) auxin efflux carriers. Flavonoids, a class of secondary plant metabolites, have been suspected to modulate auxin transport and tropic responses. Nevertheless, the identity of specific flavonoid compounds involved and their molecular function and targets in vivo are essentially unknown. Here we show that the root elongation zone of agravitropic pin2/eir1/wav6/agr1 has an altered pattern and amount of flavonol glycosides. Application of nanomolar concentrations of flavonols to pin2 roots is sufficient to partially restore root gravitropism. By employing a quantitative cell biological approach, we demonstrate that flavonoids partially restore the formation of lateral auxin gradients in the absence of PIN2. Chemical complementation by flavonoids correlates with an asymmetric distribution of the PIN1 protein. pin2 complementation probably does not result from inhibition of auxin efflux, as supply of the auxin transport inhibitor N-1-naphthylphthalamic acid failed to restore pin2 gravitropism. We propose that flavonoids promote asymmetric PIN shifts during gravity stimulation, thus redirecting basipetal auxin streams necessary for root bending.
Figures
Similar articles
-
Complex regulation of Arabidopsis AGR1/PIN2-mediated root gravitropic response and basipetal auxin transport by cantharidin-sensitive protein phosphatases.Plant J. 2005 Apr;42(2):188-200. doi: 10.1111/j.1365-313X.2005.02369.x. Plant J. 2005. PMID: 15807782
-
Narciclasine modulates polar auxin transport in Arabidopsis roots.J Plant Physiol. 2011 Jul 15;168(11):1149-56. doi: 10.1016/j.jplph.2011.01.025. Epub 2011 Apr 20. J Plant Physiol. 2011. PMID: 21511360
-
Root gravitropism and root hair development constitute coupled developmental responses regulated by auxin homeostasis in the Arabidopsis root apex.New Phytol. 2013 Mar;197(4):1130-1141. doi: 10.1111/nph.12092. Epub 2012 Dec 18. New Phytol. 2013. PMID: 23252740
-
Auxin and Root Gravitropism: Addressing Basic Cellular Processes by Exploiting a Defined Growth Response.Int J Mol Sci. 2021 Mar 9;22(5):2749. doi: 10.3390/ijms22052749. Int J Mol Sci. 2021. PMID: 33803128 Free PMC article. Review.
-
Spatiotemporal asymmetric auxin distribution: a means to coordinate plant development.Cell Mol Life Sci. 2006 Dec;63(23):2738-54. doi: 10.1007/s00018-006-6116-5. Cell Mol Life Sci. 2006. PMID: 17013565 Free PMC article. Review.
Cited by
-
Gravitropic response induced by coumarin: evidences of ROS distribution involvement.Plant Signal Behav. 2013 Feb;8(2):e23156. doi: 10.4161/psb.23156. Epub 2013 Jan 8. Plant Signal Behav. 2013. PMID: 23299434 Free PMC article.
-
Integrated metabolomic and transcriptomic study unveils the gene regulatory mechanisms of sugarcane growth promotion during interaction with an endophytic nitrogen-fixing bacteria.BMC Plant Biol. 2023 Jan 24;23(1):54. doi: 10.1186/s12870-023-04065-6. BMC Plant Biol. 2023. PMID: 36694111 Free PMC article.
-
The grapevine R2R3-MYB transcription factor VvMYBF1 regulates flavonol synthesis in developing grape berries.Plant Physiol. 2009 Nov;151(3):1513-30. doi: 10.1104/pp.109.142059. Epub 2009 Sep 9. Plant Physiol. 2009. PMID: 19741049 Free PMC article.
-
GmPIN-dependent polar auxin transport is involved in soybean nodule development.Plant Cell. 2021 Sep 24;33(9):2981-3003. doi: 10.1093/plcell/koab183. Plant Cell. 2021. PMID: 34240197 Free PMC article.
-
Plant Cell Cancer: May Natural Phenolic Compounds Prevent Onset and Development of Plant Cell Malignancy? A Literature Review.Molecules. 2016 Aug 23;21(9):1104. doi: 10.3390/molecules21091104. Molecules. 2016. PMID: 27563858 Free PMC article. Review.
References
-
- Muday, G. K. (2001) J. Plant Growth Regul. 20 226-243 - PubMed
-
- Moore, I. (2002) Curr. Biol. 12 R452-454 - PubMed
-
- Vieten, A., Sauer, M., Brewer, P. B., and Friml, J. (2007) Trends Plant Sci. 12 160-168 - PubMed
-
- Wisniewska, J., Xu, J., Seifertova, D., Brewer, P. B., Ruzicka, K., Blilou, I., Rouquie, D., Benkova, E., Scheres, B., and Friml, J. (2006) Science 312 883. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

