Synthesis, acoustic stability, and pharmacologic activities of papaverine-loaded echogenic liposomes for ultrasound controlled drug delivery
- PMID: 18720194
- PMCID: PMC2846712
- DOI: 10.1080/08982100802354558
Synthesis, acoustic stability, and pharmacologic activities of papaverine-loaded echogenic liposomes for ultrasound controlled drug delivery
Abstract
Background: development of encapsulated therapeutics that could be released upon ultrasound exposure has strong implications for enhancing drug effects at the target site. We have developed echogenic liposomes (ELIP) suitable for ultrasound imaging of blood flow and ultrasound-mediated intravascular drug release. Papaverine was chosen as the test drug because its clinical application requires high concentration in the target vascular bed but low concentration in the systemic circulation.
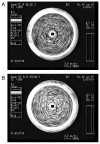
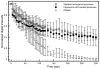
Methods: the procedure for preparation of standard ELIP was modified by including Papaverine hydrochloride in the lipid hydration solution, followed by three freeze-thaw cycles to increase encapsulation of the drug. Sizing and encapsulation pharmacokinetics were performed using a Coulter counter and a phosphodiesterase activity assay. Stability of Papaverine-loaded ELIP (PELIP) was monitored with a clinical diagnostic ultrasound scanner equipped with a linear array transducer at a center frequency of 4.5 MHz by assessing the mean digital intensity within a region of interest over time. The stability of PELIP was compared to those of standard ELIP and Optison.
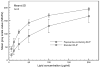
Results: relative to standard ELIP, PELIP were larger (median diameter = 1.88 +/- 0.10 microm for PELIP vs 1.08 +/- 0.15 microm for ELIP) and had lower Mean Gray Scale Values (MGSV) (92 +/- 24.8 for PELIP compared to 142.3 +/- 10.7 for ELIP at lipid concentrations of 50 microg/ml). The maximum loading efficiency and mean encapsulated concentration were 24% +/- 7% and 2.1 +/- 0.7 mg/ml, respectively. Papaverine retained its phosphodiesterase inhibitory activity when associated with PELIP. Furthermore, a fraction of this activity remained latent until released by dissolution of liposomal membranes with detergent. The stability of both PELIP and standard ELIP were similar, but both are greater than that of Optison.
Conclusions: our results suggest that PELIP have desirable physical, biochemical, biological, and acoustic characteristics for potential in vivo administration and ultrasound-controlled drug delivery.
Figures


Similar articles
-
Ultrasound-mediated release of hydrophilic and lipophilic agents from echogenic liposomes.J Ultrasound Med. 2008 Nov;27(11):1597-606. doi: 10.7863/jum.2008.27.11.1597. J Ultrasound Med. 2008. PMID: 18946099 Free PMC article.
-
Ultrasound-triggered release of recombinant tissue-type plasminogen activator from echogenic liposomes.Ultrasound Med Biol. 2010 Jan;36(1):145-57. doi: 10.1016/j.ultrasmedbio.2009.08.009. Ultrasound Med Biol. 2010. PMID: 19900755 Free PMC article.
-
Plasmin-loaded echogenic liposomes for ultrasound-mediated thrombolysis.Transl Stroke Res. 2015 Feb;6(1):78-87. doi: 10.1007/s12975-014-0376-4. Epub 2014 Nov 20. Transl Stroke Res. 2015. PMID: 25411015 Free PMC article.
-
Ultrasound, liposomes, and drug delivery: principles for using ultrasound to control the release of drugs from liposomes.Chem Phys Lipids. 2009 Nov;162(1-2):1-16. doi: 10.1016/j.chemphyslip.2009.08.003. Epub 2009 Aug 22. Chem Phys Lipids. 2009. PMID: 19703435 Review.
-
Liposomes as carriers of hydrophilic small molecule drugs: strategies to enhance encapsulation and delivery.Colloids Surf B Biointerfaces. 2014 Nov 1;123:345-63. doi: 10.1016/j.colsurfb.2014.09.029. Epub 2014 Sep 22. Colloids Surf B Biointerfaces. 2014. PMID: 25280609 Review.
Cited by
-
Nitric oxide-loaded echogenic liposomes for nitric oxide delivery and inhibition of intimal hyperplasia.J Am Coll Cardiol. 2009 Aug 11;54(7):652-9. doi: 10.1016/j.jacc.2009.04.039. J Am Coll Cardiol. 2009. PMID: 19660697 Free PMC article.
-
In vitro methods to study bubble-cell interactions: Fundamentals and therapeutic applications.Biomicrofluidics. 2016 Jan 28;10(1):011501. doi: 10.1063/1.4940429. eCollection 2016 Jan. Biomicrofluidics. 2016. PMID: 26865903 Free PMC article. Review.
-
Ultrasound-mediated release of hydrophilic and lipophilic agents from echogenic liposomes.J Ultrasound Med. 2008 Nov;27(11):1597-606. doi: 10.7863/jum.2008.27.11.1597. J Ultrasound Med. 2008. PMID: 18946099 Free PMC article.
-
Ultrasound-enhanced delivery of targeted echogenic liposomes in a novel ex vivo mouse aorta model.J Control Release. 2010 Jun 15;144(3):288-95. doi: 10.1016/j.jconrel.2010.02.030. Epub 2010 Mar 2. J Control Release. 2010. PMID: 20202474 Free PMC article.
-
The impact of bubbles on measurement of drug release from echogenic liposomes.Ultrason Sonochem. 2013 Jul;20(4):1121-30. doi: 10.1016/j.ultsonch.2012.12.005. Epub 2012 Dec 29. Ultrason Sonochem. 2013. PMID: 23357288 Free PMC article.
References
-
- Alkan-Onyuksel H, Demos SM, Lanza GM, Vonesh MJ, Klegerman ME, Kane BJ, Kuszak J, McPherson DD. Development of inherently echogenic liposomes as an ultrasonic contrast agent. J Pharm Sci. 1996;85:486–90. - PubMed
-
- Allen TM. Liposomal drug formulations. Rationale for development and what we can expect for the future. Drugs. 1998;56:747–56. - PubMed
-
- Andaluz N, Tomsick TA, Tew JM, Jr, van Loveren HR, Yeh HS, Zuccarello M. Indications for endovascular therapy for refractory vasospasm after aneurysmal subarachnoid hemorrhage: Experience at the University of Cincinnati. Surg Neurol. 2002;58:131–8. - PubMed
-
- Fandino J, Kaku Y, Schuknecht B, Valavanis A, Yonekowa Y. Improvement of cerebral oxygenation patterns and metabolic validation of superselective intraarterial infusion of papaverine for the treatment of cerebral vasospasm. Journal of Neurosurgery. 1998;89:93–100. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
