Increased arachidonic acid-induced thromboxane generation impairs skeletal muscle arteriolar dilation with genetic dyslipidemia
- PMID: 18720229
- PMCID: PMC3043548
- DOI: 10.1080/10739680802308334
Increased arachidonic acid-induced thromboxane generation impairs skeletal muscle arteriolar dilation with genetic dyslipidemia
Abstract
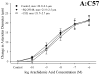
Objective: The aim of this study was to determine if arachidonic acid (AA)-induced skeletal muscle arteriolar dilation is altered with hypercholesterolemia in ApoE and low-density lipoprotein receptor (LDLR) gene deletion mice fed a normal diet. This study also determined contributors to altered AA-induced dilation between dyslipidemic mice and controls, C57/Bl/6J (C57).
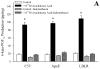
Methods: Gracilis muscle arterioles were isolated, with mechanical responses assessed following a challenge with AA under control conditions and after elements of AA metabolism pathways were inhibited. Conduit arteries from each strain were used to assess AA-induced production of PGI(2) and TxA(2).
Results: Arterioles from ApoE and LDLR exhibited a blunted dilation to AA versus C57. While responses were cyclo-oxygenase-dependent in all strains, inhibition of thromboxane synthase or blockade of PGH(2)/TxA(2) receptors improved dilation in ApoE and LDLR only. AA-induced generation of PGI(2) was comparable across strains, although TxA(2) generation was increased in ApoE and LDLR. Arteriolar reactivity to PGI(2) and TxA(2) was comparable across strains. Treatment with TEMPOL improved dilation and reduced TxA(2) production with AA in ApoE and LDLR.
Conclusions: These results suggest that AA-induced arteriolar dilation is constrained in ApoE and LDLR via an increased production of TxA(2). While partially due to elevated oxidant stress, additional mechanisms contribute that are independent of acute alterations in oxidant tone.
Figures













References
-
- American Heart Association Statistical Summary Sheets. High Blood Cholesterol and Other Lipids. 2007. http://www.americanheart.org/presenter.jhtml?identifier=3000945.
-
- Bachschmid M, Thurau S, Zou MH, Ullrich V. Endothelial cell activation by endotoxin involves superoxide/NO-mediated nitration of prostacyclin synthase and thromboxane receptor stimulation. FASEB J. 2003;17:914–6. - PubMed
-
- Bhatt DL, Steg PG, Ohman EM, Hirsch AT, Ikeda Y, Mas JL, Goto S, Liau CS, Richard AJ, Rother J, Wilson PW REACH Registry Investigators. International prevalence, recognition, and treatment of cardiovascular risk factors in outpatients with atherothrombosis. JAMA. 2006;295:180–9. - PubMed
-
- de Jongh S, Lilien MR, op’t Roodt J, Stroes ES, Bakker HD, Kastelein JJ. Early statin therapy restores endothelial function in children with familial hypercholesterolemia. J Am Coll Cardiol. 2002;40:2117–21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

