A quantitative study of spinothalamic neurons in laminae I, III, and IV in lumbar and cervical segments of the rat spinal cord
- PMID: 18720412
- PMCID: PMC2658017
- DOI: 10.1002/cne.21811
A quantitative study of spinothalamic neurons in laminae I, III, and IV in lumbar and cervical segments of the rat spinal cord
Abstract
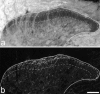
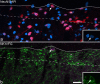
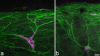
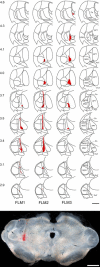
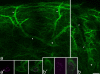
The major ascending outputs from superficial spinal dorsal horn consist of projection neurons in lamina I, together with neurons in laminae III-IV that express the neurokinin 1 receptor (NK1r) and have dendrites that enter the superficial laminae. Some neurons in each of these populations belong to the spinothalamic tract, which conveys nociceptive information via the thalamus to cortical areas involved in pain. A projection from the cervical superficial dorsal horn to the posterior triangular nucleus (PoT) has recently been identified. PoT is at the caudal end of the thalamus and was not included in injection sites in many previous retrograde tracing studies. We have injected various tracers (cholera toxin B subunit, Fluoro-Gold, and fluorescent latex microspheres) into the thalamus to estimate the number of spinothalamic neurons in each of these two populations, and to investigate their projection targets. Most lamina I and lamina III/IV NK1r-immunoreactive spinothalamic neurons in cervical and lumbar segments could be labeled from injections centered on PoT. Our results suggest that there are 90 lamina I spinothalamic neurons per side in C7 and 15 in L4 and that some of those in C7 only project to PoT. We found that 85% of the lamina III/IV NK1r-immunoreactive neurons in C6 and 17% of those in L5 belong to the spinothalamic tract, and these apparently project exclusively to the caudal thalamus, including PoT. Because PoT projects to second somatosensory and insular cortices, our results suggest that these are major targets for information conveyed by both these populations of spinothalamic neurons.
Figures




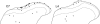


References
-
- Akintunde A, Buxton DF. Quadruple labeling of brain-stem neurons: a multiple retrograde fluorescent tracer study of axonal collateralization. J Neurosci Methods. 1992;45:15–22. - PubMed
-
- Apkarian AV, Hodge CJ. Primate spinothalamic pathways: I. A quantitative study of the cells of origin of the spinothalamic pathway. J Comp Neurol. 1989;288:447–473. - PubMed
-
- Bester H, Chapman V, Besson JM, Bernard JF. Physiological properties of the lamina I spinoparabrachial neurons in the rat. J Neurophysiol. 2000;83:2239–2259. - PubMed
-
- Bice TN, Beal JA. Quantitative and neurogenic analysis of the total population and subpopulations of neurons defined by axon projection in the superficial dorsal horn of the rat lumbar spinal cord. J Comp Neurol. 1997;388:550–564. - PubMed
-
- Brown JL, Liu H, Maggio JE, Vigna SR, Mantyh PW, Basbaum AI. Morphological characterization of substance P receptor-immunoreactive neurons in the rat spinal cord and trigeminal nucleus caudalis. J Comp Neurol. 1995;356:327–344. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

