Senescence of activated stellate cells limits liver fibrosis
- PMID: 18724938
- PMCID: PMC3073300
- DOI: 10.1016/j.cell.2008.06.049
Senescence of activated stellate cells limits liver fibrosis
Abstract
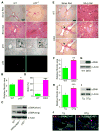
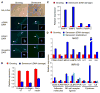
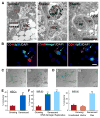
Cellular senescence acts as a potent mechanism of tumor suppression; however, its functional contribution to noncancer pathologies has not been examined. Here we show that senescent cells accumulate in murine livers treated to produce fibrosis, a precursor pathology to cirrhosis. The senescent cells are derived primarily from activated hepatic stellate cells, which initially proliferate in response to liver damage and produce the extracellular matrix deposited in the fibrotic scar. In mice lacking key senescence regulators, stellate cells continue to proliferate, leading to excessive liver fibrosis. Furthermore, senescent activated stellate cells exhibit gene expression profile consistent with cell-cycle exit, reduced secretion of extracellular matrix components, enhanced secretion of extracellular matrix-degrading enzymes, and enhanced immune surveillance. Accordingly natural killer cells preferentially kill senescent activated stellate cells in vitro and in vivo, thereby facilitating the resolution of fibrosis. Therefore, the senescence program limits the fibrogenic response to acute tissue damage.
Figures




Comment in
- Cell. 2008 October 3;135(1):190
-
Senescence: not just for tumor suppression.Cell. 2008 Aug 22;134(4):562-4. doi: 10.1016/j.cell.2008.08.003. Cell. 2008. PMID: 18724927
-
Senescence of activated stellate cells: not just early retirement.Hepatology. 2009 Mar;49(3):1045-7. doi: 10.1002/hep.22832. Hepatology. 2009. PMID: 19242970 No abstract available.
References
-
- Bartkova J, Rezaei N, Liontos M, Karakaidos P, Kletsas D, Issaeva N, Vassiliou LV, Kolettas E, Niforou K, Zoumpourlis VC, et al. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature. 2006;444:633–637. - PubMed
-
- Berenguer M, Ferrell L, Watson J, Prieto M, Kim M, Rayon M, Cordoba J, Herola A, Ascher N, Mir J, et al. HCV-related fibrosis progression following liver transplantation: increase in recent years. J Hepatol. 2000;32:673–684. - PubMed
-
- Braig M, Lee S, Loddenkemper C, Rudolph C, Peters AH, Schlegelberger B, Stein H, Dorken B, Jenuwein T, Schmitt CA. Oncogene-induced senescence as an initial barrier in lymphoma development. Nature. 2005;436:660–665. - PubMed
-
- Campisi J, d’Adda di Fagagna F. Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol. 2007;8:729–740. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

