Trans-Golgi network and endosome dynamics connect ceramide homeostasis with regulation of the unfolded protein response and TOR signaling in yeast
- PMID: 18753406
- PMCID: PMC2575146
- DOI: 10.1091/mbc.e08-04-0426
Trans-Golgi network and endosome dynamics connect ceramide homeostasis with regulation of the unfolded protein response and TOR signaling in yeast
Abstract
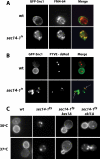
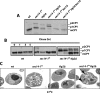
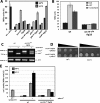
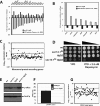
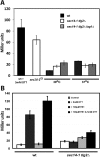
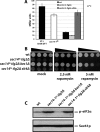
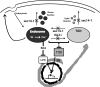
Synthetic genetic array analyses identify powerful genetic interactions between a thermosensitive allele (sec14-1(ts)) of the structural gene for the major yeast phosphatidylinositol transfer protein (SEC14) and a structural gene deletion allele (tlg2Delta) for the Tlg2 target membrane-soluble N-ethylmaleimide-sensitive factor attachment protein receptor. The data further demonstrate Sec14 is required for proper trans-Golgi network (TGN)/endosomal dynamics in yeast. Paradoxically, combinatorial depletion of Sec14 and Tlg2 activities elicits trafficking defects from the endoplasmic reticulum, and these defects are accompanied by compromise of the unfolded protein response (UPR). UPR failure occurs downstream of Hac1 mRNA splicing, and it is further accompanied by defects in TOR signaling. The data link TGN/endosomal dynamics with ceramide homeostasis, UPR activity, and TOR signaling in yeast, and they identify the Sit4 protein phosphatase as a primary conduit through which ceramides link to the UPR. We suggest combinatorial Sec14/Tlg2 dysfunction evokes inappropriate turnover of complex sphingolipids in endosomes. One result of this turnover is potentiation of ceramide-activated phosphatase-mediated down-regulation of the UPR. These results provide new insight into Sec14 function, and they emphasize the TGN/endosomal system as a central hub for homeostatic regulation in eukaryotes.
Figures



Similar articles
-
Sphingolipid metabolism in trans-golgi/endosomal membranes and the regulation of intracellular homeostatic processes in eukaryotic cells.Adv Enzyme Regul. 2010;50(1):339-48. doi: 10.1016/j.advenzreg.2009.12.004. Epub 2009 Dec 23. Adv Enzyme Regul. 2010. PMID: 20005891 Review. No abstract available.
-
Phospholipid transfer protein Sec14 is required for trafficking from endosomes and regulates distinct trans-Golgi export pathways.J Biol Chem. 2009 Mar 13;284(11):7364-75. doi: 10.1074/jbc.M808732200. Epub 2009 Jan 6. J Biol Chem. 2009. PMID: 19129178 Free PMC article.
-
The functional relationship between the Cdc50p-Drs2p putative aminophospholipid translocase and the Arf GAP Gcs1p in vesicle formation in the retrieval pathway from yeast early endosomes to the TGN.Cell Struct Funct. 2006;31(2):87-108. doi: 10.1247/csf.06021. Epub 2006 Oct 23. Cell Struct Funct. 2006. PMID: 17062999
-
Role of the unfolded protein response pathway in regulation of INO1 and in the sec14 bypass mechanism in Saccharomyces cerevisiae.Genetics. 2002 Sep;162(1):29-43. doi: 10.1093/genetics/162.1.29. Genetics. 2002. PMID: 12242221 Free PMC article.
-
CERT and intracellular trafficking of ceramide.Biochim Biophys Acta. 2007 Jun;1771(6):644-53. doi: 10.1016/j.bbalip.2007.01.009. Epub 2007 Jan 23. Biochim Biophys Acta. 2007. PMID: 17314061 Review.
Cited by
-
Valproate Induces the Unfolded Protein Response by Increasing Ceramide Levels.J Biol Chem. 2016 Oct 14;291(42):22253-22261. doi: 10.1074/jbc.M116.752634. Epub 2016 Sep 1. J Biol Chem. 2016. PMID: 27590340 Free PMC article.
-
The retrograde response: a conserved compensatory reaction to damage from within and from without.Prog Mol Biol Transl Sci. 2014;127:133-54. doi: 10.1016/B978-0-12-394625-6.00005-2. Prog Mol Biol Transl Sci. 2014. PMID: 25149216 Free PMC article. Review.
-
Role for Lipid Droplet Biogenesis and Microlipophagy in Adaptation to Lipid Imbalance in Yeast.Dev Cell. 2015 Dec 7;35(5):584-599. doi: 10.1016/j.devcel.2015.11.010. Dev Cell. 2015. PMID: 26651293 Free PMC article.
-
Aggregation of α-synuclein in S. cerevisiae is associated with defects in endosomal trafficking and phospholipid biosynthesis.J Mol Neurosci. 2011 Mar;43(3):391-405. doi: 10.1007/s12031-010-9455-5. Epub 2010 Oct 2. J Mol Neurosci. 2011. PMID: 20890676 Free PMC article.
-
Vesicle trafficking from a lipid perspective: Lipid regulation of exocytosis in Saccharomyces cerevisiae.Cell Logist. 2012 Jul 1;2(3):151-160. doi: 10.4161/cl.20490. Cell Logist. 2012. PMID: 23181198 Free PMC article.
References
-
- Abeliovich H., Grote E., Novick P., Ferro-Novick S. Tlg2p, a yeast syntaxin homolog that resides on the Golgi and endocytic structures. J. Biol. Chem. 1998;273:11719–11727. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

