Amorphous calcium phosphate is a major component of the forming fin bones of zebrafish: Indications for an amorphous precursor phase
- PMID: 18753619
- PMCID: PMC2529085
- DOI: 10.1073/pnas.0803354105
Amorphous calcium phosphate is a major component of the forming fin bones of zebrafish: Indications for an amorphous precursor phase
Abstract
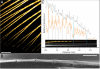
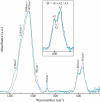
A fundamental question in biomineralization is the nature of the first-formed mineral phase. In vertebrate bone formation, this issue has been the subject of a long-standing controversy. We address this key issue using the continuously growing fin bony rays of the Tuebingen long-fin zebrafish as a model for bone mineralization. Employing high-resolution scanning and transmission electron microscopy imaging, electron diffraction, and elemental analysis, we demonstrate the presence of an abundant amorphous calcium phosphate phase in the newly formed fin bones. The extracted amorphous mineral particles crystallize with time, and mineral crystallinity increases during bone maturation. Based on these findings, we propose that this amorphous calcium phosphate phase may be a precursor phase that later transforms into the mature crystalline mineral.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Towe KM, Lowenstam HA. Ultrastructure and development of iron mineralization in the radular teeth of Cryptochiton stelleri (Mollusca) J Ultrastructure Res. 1967;17:1–13. - PubMed
-
- Lowenstam HA, Weiner S. Transformation of amorphous calcium phosphate to crystalline dahillite in the radular teeth of chitons. Science. 1985;227:51–53. - PubMed
-
- Beniash E, Aizenberg J, Addadi L, Weiner S. Amorphous calcium carbonate transforms into calcite during sea-urchin larval spicule growth. Proc R Soc London Ser B. 1997;64:461–465.
-
- Hasse B, et al. Calcium carbonate modifications in the mineralized shell of the freshwater snail Biomphalaria glabrata. Chem Eur J. 2000;6:3679–3685. - PubMed
-
- Weiss IM, Tuross N, Addadi L, Weiner S. Mollusk larval shell formation: amorphous calcium carbonate is a precursor for aragonite. J Exp Zool. 2002;293:478–491. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

