Leydig cells express follicle-stimulating hormone receptors in African catfish
- PMID: 18755797
- PMCID: PMC2732288
- DOI: 10.1210/en.2008-0447
Leydig cells express follicle-stimulating hormone receptors in African catfish
Abstract
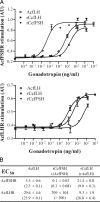
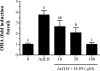
This report aimed to establish, using African catfish, Clarias gariepinus, as model species, a basis for understanding a well-known, although not yet clarified, feature of male fish reproductive physiology: the strong steroidogenic activity of FSHs. Assays with gonadotropin receptor-expressing cell lines showed that FSH activated its cognate receptor (FSHR) with an at least 1000-fold lower EC50 than when challenging the LH receptor (LHR), whereas LH stimulated both receptors with similar EC50s. In androgen release bioassays, FSH elicited a significant response at lower concentrations than those required to cross-activate of the LHR, indicating that FSH stimulated steroid release via FSHR-dependent mechanisms. LHR/FSHR-mediated stimulation of androgen release was completely abolished by H-89, a specific protein kinase A inhibitor, pointing to the cAMP/protein kinase A pathway as the main route for both LH- and FSH-stimulated steroid release. Localization studies showed that intratubular Sertoli cells express FSHR mRNA, whereas, as reported for the first time in a vertebrate, catfish Leydig cells express both LHR and FSHR mRNA. Testicular FSHR and LHR mRNA expression increased gradually during pubertal development. FSHR, but not LHR, transcript levels continued to rise between completion of the first wave of spermatogenesis at about 7 months and full maturity at about 12 months of age, which was associated with a previously recorded approximately 3-fold increase in the steroid production capacity per unit testis weight. Taken together, our data strongly suggest that the steroidogenic potency of FSH can be explained by its direct trophic action on FSHR-expressing Leydig cells.
Figures




Similar articles
-
Studies in zebrafish reveal unusual cellular expression patterns of gonadotropin receptor messenger ribonucleic acids in the testis and unexpected functional differentiation of the gonadotropins.Endocrinology. 2010 May;151(5):2349-60. doi: 10.1210/en.2009-1227. Epub 2010 Mar 22. Endocrinology. 2010. PMID: 20308533 Free PMC article.
-
An activated human follicle-stimulating hormone (FSH) receptor stimulates FSH-like activity in gonadotropin-deficient transgenic mice.Mol Endocrinol. 2002 Nov;16(11):2582-91. doi: 10.1210/me.2002-0032. Mol Endocrinol. 2002. PMID: 12403847
-
Follicle-stimulating hormone and luteinizing hormone mediate the androgenic pathway in Leydig cells of an evolutionary advanced teleost.Biol Reprod. 2012 Aug 9;87(2):35. doi: 10.1095/biolreprod.112.100784. Print 2012 Aug. Biol Reprod. 2012. PMID: 22649073
-
Gonadotropin receptors and the control of gonadal steroidogenesis: physiology and pathology.Baillieres Clin Endocrinol Metab. 1998 Apr;12(1):35-66. doi: 10.1016/s0950-351x(98)80444-8. Baillieres Clin Endocrinol Metab. 1998. PMID: 9890061 Review.
-
Gonadotropins, their receptors, and the regulation of testicular functions in fish.Comp Biochem Physiol B Biochem Mol Biol. 2001 Jun;129(2-3):407-17. doi: 10.1016/s1096-4959(01)00339-6. Comp Biochem Physiol B Biochem Mol Biol. 2001. PMID: 11399475 Review.
Cited by
-
Post-spawning feed deprivation effects on testicular and ovarian maturation in the neotropical cichlid fish Cichlasoma dimerus.Fish Physiol Biochem. 2021 Oct;47(5):1527-1540. doi: 10.1007/s10695-021-00993-y. Epub 2021 Aug 3. Fish Physiol Biochem. 2021. PMID: 34341877
-
Chronic exposure to aquacultural stressors affects pituitary-testis axis in the Mozambique tilapia Oreochromis mossambicus.Fish Physiol Biochem. 2022 Apr;48(2):437-448. doi: 10.1007/s10695-022-01061-9. Epub 2022 Feb 24. Fish Physiol Biochem. 2022. PMID: 35201519
-
Germ-line activation of the luteinizing hormone receptor directly drives spermiogenesis in a nonmammalian vertebrate.Proc Natl Acad Sci U S A. 2014 Jan 28;111(4):1427-32. doi: 10.1073/pnas.1317838111. Epub 2014 Jan 13. Proc Natl Acad Sci U S A. 2014. PMID: 24474769 Free PMC article.
-
Identification of conserved genes triggering puberty in European sea bass males (Dicentrarchus labrax) by microarray expression profiling.BMC Genomics. 2017 Jun 5;18(1):441. doi: 10.1186/s12864-017-3823-2. BMC Genomics. 2017. PMID: 28583077 Free PMC article.
-
The Potential Role of Gonadotropic Hormones and Their Receptors in Sex Differentiation of Nile Tilapia, Oreochromis niloticus.Int J Mol Sci. 2025 Jun 4;26(11):5376. doi: 10.3390/ijms26115376. Int J Mol Sci. 2025. PMID: 40508194 Free PMC article.
References
-
- McLachlan RI, Wreford NG, O'Donnell L, De Kretser DM, Robertson DM 1996 The endocrine regulation of spermatogenesis: independent roles for testosterone and FSH. J Endocrinol 14:1–9 - PubMed
-
- Vischer HF, Granneman JCM, Linskens MHK, Schulz RW, Bogerd J 2003 Both recombinant African catfish LH and FSH are able to activate the African catfish FSH receptor. J Mol Endocrinol 31:133–140 - PubMed
-
- Pierce JG, Parsons TF 1981 Glycoprotein hormones: structure and function. Ann Rev Biochem 50:465–495 - PubMed
-
- Jia XC, Oikawa M, Bo M, Tanaka T, Ny T, Boime I, Hsueh AJ 1991 Expression of human luteinizing hormone (LH) receptor: interaction with LH and chorionic gonadotropin from human but not equine, rat, and ovine species. Mol Endocrinol 5:759–768 - PubMed
-
- Tilly JL, Aihara T, Nishimori K, Jia XC, Billig H, Kowalsky KI, Perlas EA, Hsueh AJ 1992 Expression of human follicle-stimulating hormone receptor: species-specific ligand binding, signal transduction, and identification of multiple ovarian messenger ribonucleic acid transcripts. Endocrinology 131:799–806 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

