NeuroD1 and Mash1 temporally regulate GnRH receptor gene expression in immortalized mouse gonadotrope cells
- PMID: 18760324
- PMCID: PMC2640340
- DOI: 10.1016/j.mce.2008.07.017
NeuroD1 and Mash1 temporally regulate GnRH receptor gene expression in immortalized mouse gonadotrope cells
Abstract
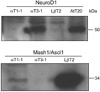
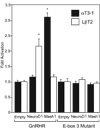
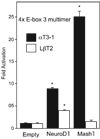
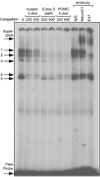
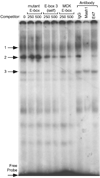
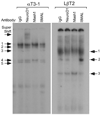
Accurate spatial and temporal expression of gonadotrope-specific genes, such as the gonadotropin-releasing hormone receptor (GnRHR) gene, is critical for gonadotrope maturation. Herein, we show that a specific E-box in the mouse GnRHR promoter binds two group A basic-helix-loop-helix (bHLH) transcription factors. Mutation of this E-box decreases expression in mouse gonadotrope-derived alphaT3-1 and LbetaT2 cell lines. Microarray and western blots show that the bHLH transcription factor NeuroD1 is strongly expressed in the gonadotrope progenitor, alphaT3-1, whereas Mash1 is strongly expressed in the more mature gonadotrope, LbetaT2. Over-expression of NeuroD1 or Mash1 increases expression of the GnRHR gene or a multimer of the E-box and this increase is lost upon mutation of the E-box. Electrophoretic mobility shift assays reveal that the GnRHR E-box binds NeuroD1 from alphaT3-1 cells, but binds Mash1 from LbetaT2 cells. The sequential binding of different members of the group A bHLH transcription factor family to mouse GnRHR E-box 3 as the gonadotrope differentiates may represent a mechanism necessary for proper spatial and temporal expression of the GnRHR during gonadotrope development.
Figures

References
-
- Alarid ET, Windle JJ, Whyte DB, Mellon PL. Immortalization of pituitary cells at discrete stages of development by directed oncogenesis in transgenic mice. Development. 1996;122:3319–3329. - PubMed
-
- Albarracin CT, Kaiser UB, Chin WW. Isolation and characterization of the 5'-flanking region of the mouse gonadotropin-releasing hormone receptor gene. Endocrinology. 1994;135:2300–2306. - PubMed
-
- Duval DL, Nelson SE, Clay CM. The tripartite basal enhancer of the gonadotropin-releasing hormone (GnRH) receptor gene promoter regulates cell-specific expression through a novel GnRH receptor activating sequence. Mol. Endocrinol. 1997;11:1814–1821. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

