Neuregulin-1/ErbB signaling serves distinct functions in myelination of the peripheral and central nervous system
- PMID: 18760695
- PMCID: PMC2628490
- DOI: 10.1016/j.neuron.2008.06.028
Neuregulin-1/ErbB signaling serves distinct functions in myelination of the peripheral and central nervous system
Abstract
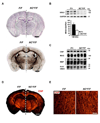
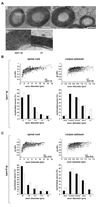
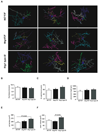
Understanding the control of myelin formation by oligodendrocytes is essential for treating demyelinating diseases. Neuregulin-1 (NRG1) type III, an EGF-like growth factor, is essential for myelination in the PNS. It is thus thought that NRG1/ErbB signaling also regulates CNS myelination, a view suggested by in vitro studies and the overexpression of dominant-negative ErbB receptors. To directly test this hypothesis, we generated a series of conditional null mutants that completely lack NRG1 beginning at different stages of neural development. Unexpectedly, these mice assemble normal amounts of myelin. In addition, double mutants lacking oligodendroglial ErbB3 and ErbB4 become myelinated in the absence of any stimulation by neuregulins. In contrast, a significant hypermyelination is achieved by transgenic overexpression of NRG1 type I or NRG1 type III. Thus, NRG1/ErbB signaling is markedly different between Schwann cells and oligodendrocytes that have evolved an NRG/ErbB-independent mechanism of myelination control.
Figures





References
-
- Adlkofer K, Lai C. Role of neuregulins in glial cell development. Glia. 2000;29:104–111. - PubMed
-
- Aguirre A, Dupree JL, Mangin JM, Gallo V. A functional role for EGFR signaling in myelination and remyelination. Nat Neurosci. 2007;10:990–1002. - PubMed
-
- Bartzokis G. Schizophrenia: breakdown in the well-regulated lifelong process of brain development and maturation. Neuropsychopharmacology. 2002;27:672–683. - PubMed
-
- Busfield SJ, Michnick DA, Chickering TW, Revett TL, Ma J, Woolf EA, Comrack CA, Dussault BJ, Woolf J, Goodearl AD, Gearing DP. Characterization of a neuregulin-related gene, Don-1, that is highly expressed in restricted regions of the cerebellum and hippocampus. Mol Cell Biol. 1997;17:4007–4014. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

