Subversion of complement by hematophagous parasites
- PMID: 18762211
- PMCID: PMC2642905
- DOI: 10.1016/j.dci.2008.07.010
Subversion of complement by hematophagous parasites
Abstract
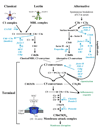
The complement system is a crucial part of innate and adaptive immunity which exerts a significant evolutionary pressure on pathogens. It has selected for those pathogens, mainly microorganisms but also parasites, that have evolved countermeasures. The characterization of how pathogens evade complement attack is a rapidly developing field of current research. In recent years, multiple complement evasion strategies have been characterized. In this review, we focus on complement escape mechanisms expressed by hematophagous parasites, a heterogeneous group of metazoan parasites that share the property of ingesting the whole blood of their host. Complement inhibition is crucial for parasite survival within the host tissue or to facilitate blood feeding. Finally, complement inhibition by hematophagous parasites may also contribute to their success as pathogen vectors.
Figures


References
-
- Nonaka M, Yoshizaki F. Evolution of the complement system. Mol Immunol. 2004;40(12):897–902. - PubMed
-
- Rother K, Till GO, Hänsch GM. The Complement System. Berlin: Springer; 1998.
-
- Houle J, Hoffmann E. Evidence for restriction of the ability of complement to lyse homologous erythrocytes. J Immunol. 1984;133(3):1444–1452. - PubMed
-
- Meri S, Jarva H. Complement regulation. Vol. 74. Vox Sang; 1998. pp. 291–302. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

