Commensal-dependent expression of IL-25 regulates the IL-23-IL-17 axis in the intestine
- PMID: 18762568
- PMCID: PMC2556798
- DOI: 10.1084/jem.20080720
Commensal-dependent expression of IL-25 regulates the IL-23-IL-17 axis in the intestine
Abstract
Alterations in the composition of intestinal commensal bacteria are associated with enhanced susceptibility to multiple inflammatory diseases, including those conditions associated with interleukin (IL)-17-producing CD4(+) T helper (Th17) cells. However, the relationship between commensal bacteria and the expression of proinflammatory cytokines remains unclear. Using germ-free mice, we show that the frequency of Th17 cells in the large intestine is significantly elevated in the absence of commensal bacteria. Commensal-dependent expression of the IL-17 family member IL-25 (IL-17E) by intestinal epithelial cells limits the expansion of Th17 cells in the intestine by inhibiting expression of macrophage-derived IL-23. We propose that acquisition of, or alterations in, commensal bacteria influences intestinal immune homeostasis via direct regulation of the IL-25-IL-23-IL-17 axis.
Figures

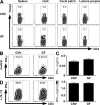
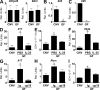

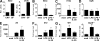
References
-
- Hooper, L.V., and J.I. Gordon. 2001. Commensal host-bacterial relationships in the gut. Science. 292:1115–1118. - PubMed
-
- Macpherson, A.J., and N.L. Harris. 2004. Interactions between commensal intestinal bacteria and the immune system. Nat. Rev. Immunol. 4:478–485. - PubMed
-
- Ley, R.E., D.A. Peterson, and J.I. Gordon. 2006. Ecological and evolutionary forces shaping microbial diversity in the human intestine. Cell. 124:837–848. - PubMed
-
- Strober, W., I.J. Fuss, and R.S. Blumberg. 2002. The immunology of mucosal models of inflammation. Annu. Rev. Immunol. 20:495–549. - PubMed
-
- Sellon, R.K., S. Tonkonogy, M. Schultz, L.A. Dieleman, W. Grenther, E. Balish, D.M. Rennick, and R.B. Sartor. 1998. Resident enteric bacteria are necessary for development of spontaneous colitis and immune system activation in interleukin-10-deficient mice. Infect. Immun. 66:5224–5231. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 AI074878/AI/NIAID NIH HHS/United States
- F32-AI72943/AI/NIAID NIH HHS/United States
- AI37108/AI/NIAID NIH HHS/United States
- T32 AI007532/AI/NIAID NIH HHS/United States
- T32 CA009140/CA/NCI NIH HHS/United States
- T32-AI007532-08/AI/NIAID NIH HHS/United States
- AI74878/AI/NIAID NIH HHS/United States
- F31 GM082187/GM/NIGMS NIH HHS/United States
- R01 AI061570/AI/NIAID NIH HHS/United States
- F31-GM82187/GM/NIGMS NIH HHS/United States
- R37 AI037108/AI/NIAID NIH HHS/United States
- T32-CA09140-30/CA/NCI NIH HHS/United States
- R01 AI037108/AI/NIAID NIH HHS/United States
- F32 AI072943/AI/NIAID NIH HHS/United States
- AI61570/AI/NIAID NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

