Role of N-terminal amino acids in the potency of anthrax lethal factor
- PMID: 18769623
- PMCID: PMC2518864
- DOI: 10.1371/journal.pone.0003130
Role of N-terminal amino acids in the potency of anthrax lethal factor
Abstract
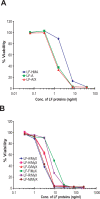
Anthrax lethal factor (LF) is a Zn(+2)-dependent metalloprotease that cleaves several MAPK kinases and is responsible for the lethality of anthrax lethal toxin (LT). We observed that a recombinant LF (LF-HMA) which differs from wild type LF (LF-A) by the addition of two residues (His-Met) to the native Ala (A) terminus as a result of cloning manipulations has 3-fold lower potency toward cultured cells and experimental animals. We hypothesized that the "N-end rule", which relates the half-life of proteins in cells to the identity of their N-terminal residue, might be operative in the case of LF, so that the N-terminal residue of LF would determine the cytosolic stability and thereby the potency of LF. Mutational studies that replaced the native N-terminal residue of LF with known N-end rule stabilizing or destabilizing residues confirmed that the N-terminal residue plays a significant role in determining the potency of LT for cultured cells and experimental animals. The fact that a commercially-available LF preparation (LF-HMA) that is widely used in basic research studies and for evaluation of vaccines and therapeutics is 3-fold less potent than native LF (LF-A) should be considered when comparing published studies and in the design of future experiments.
Conflict of interest statement
Figures


References
-
- Mourez M. Anthrax toxins. Rev Physiol Biochem Pharmacol. 2004;152:135–164. - PubMed
-
- Leppla SH. Bacillus anthracis toxins. In: Alouf JE, Popoff MR, editors. The Comprehensive Sourcebook of Bacterial Protein Toxins. Burlington, MA: Academic Press; 2006. pp. 323–347.
-
- Duesbery NS, Webb CP, Leppla SH, Gordon VM, Klimpel KR, et al. Proteolytic inactivation of MAP-kinase-kinase by anthrax lethal factor. Science. 1998;280:734–737. - PubMed
-
- Pellizzari R, Guidi-Rontani C, Vitale G, Mock M, Montecucco C. Lethal factor of Bacillus anthracis cleaves the N-terminus of MAPKKs: analysis of the intracellular consequences in macrophages. Int J Med Microbiol. 2000;290:421–427. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

