Formation of the food vacuole in Plasmodium falciparum: a potential role for the 19 kDa fragment of merozoite surface protein 1 (MSP1(19))
- PMID: 18769730
- PMCID: PMC2518119
- DOI: 10.1371/journal.pone.0003085
Formation of the food vacuole in Plasmodium falciparum: a potential role for the 19 kDa fragment of merozoite surface protein 1 (MSP1(19))
Abstract
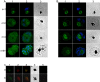
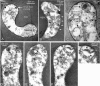
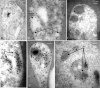
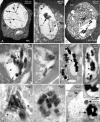
Plasmodium falciparum Merozoite Surface Protein 1 (MSP1) is synthesized during schizogony as a 195-kDa precursor that is processed into four fragments on the parasite surface. Following a second proteolytic cleavage during merozoite invasion of the red blood cell, most of the protein is shed from the surface except for the C-terminal 19-kDa fragment (MSP1(19)), which is still attached to the merozoite via its GPI-anchor. We have examined the fate of MSP1(19) during the parasite's subsequent intracellular development using immunochemical analysis of metabolically labeled MSP1(19), fluorescence imaging, and immuno-electronmicroscopy. Our data show that MSP1(19) remains intact and persists to the end of the intracellular cycle. This protein is the first marker for the biogenesis of the food vacuole; it is rapidly endocytosed into small vacuoles in the ring stage, which coalesce to form the single food vacuole containing hemozoin, and persists into the discarded residual body. The food vacuole is marked by the presence of both MSP1(19) and the chloroquine resistance transporter (CRT) as components of the vacuolar membrane. Newly synthesized MSP1 is excluded from the vacuole. This behavior indicates that MSP1(19) does not simply follow a classical lysosome-like clearance pathway, instead, it may play a significant role in the biogenesis and function of the food vacuole throughout the intra-erythrocytic phase.
Conflict of interest statement
Figures



Similar articles
-
Plasmodium falciparum 19-kilodalton merozoite surface protein 1 (MSP1)-specific antibodies that interfere with parasite growth in vitro can inhibit MSP1 processing, merozoite invasion, and intracellular parasite development.Infect Immun. 2012 Mar;80(3):1280-7. doi: 10.1128/IAI.05887-11. Epub 2011 Dec 27. Infect Immun. 2012. PMID: 22202121 Free PMC article.
-
Processing of the Plasmodium falciparum major merozoite surface protein-1: identification of a 33-kilodalton secondary processing product which is shed prior to erythrocyte invasion.Mol Biochem Parasitol. 1991 Nov;49(1):35-44. doi: 10.1016/0166-6851(91)90128-s. Mol Biochem Parasitol. 1991. PMID: 1723148
-
Malaria parasite-inhibitory antibody epitopes on Plasmodium falciparum merozoite surface protein-1(19) mapped by TROSY NMR.Mol Biochem Parasitol. 2004 Nov;138(1):29-36. doi: 10.1016/j.molbiopara.2004.06.014. Mol Biochem Parasitol. 2004. PMID: 15500913
-
The carboxy-terminus of merozoite surface protein 1: structure, specific antibodies and immunity to malaria.Parasitology. 2009 Oct;136(12):1445-56. doi: 10.1017/S0031182009990515. Epub 2009 Jul 23. Parasitology. 2009. PMID: 19627632 Review.
-
A malaria merozoite surface protein (MSP1)-structure, processing and function.Mem Inst Oswaldo Cruz. 1992;87 Suppl 3:37-42. doi: 10.1590/s0074-02761992000700004. Mem Inst Oswaldo Cruz. 1992. PMID: 1343716 Review.
Cited by
-
Oriented nucleation of hemozoin at the digestive vacuole membrane in Plasmodium falciparum.Proc Natl Acad Sci U S A. 2012 Jul 10;109(28):11188-93. doi: 10.1073/pnas.1118120109. Epub 2012 Jun 28. Proc Natl Acad Sci U S A. 2012. PMID: 22745164 Free PMC article.
-
The Digestive Vacuole of the Malaria Parasite: A Specialized Lysosome.Pathogens. 2024 Feb 20;13(3):182. doi: 10.3390/pathogens13030182. Pathogens. 2024. PMID: 38535526 Free PMC article. Review.
-
Immunomic Identification of Malaria Antigens Associated With Protection in Mice.Mol Cell Proteomics. 2019 May;18(5):837-853. doi: 10.1074/mcp.RA118.000997. Epub 2019 Feb 4. Mol Cell Proteomics. 2019. PMID: 30718293 Free PMC article.
-
Quantitative time-course profiling of parasite and host cell proteins in the human malaria parasite Plasmodium falciparum.Mol Cell Proteomics. 2011 Aug;10(8):M110.006411. doi: 10.1074/mcp.M110.006411. Epub 2011 May 10. Mol Cell Proteomics. 2011. PMID: 21558492 Free PMC article.
-
Merozoite surface proteins in red blood cell invasion, immunity and vaccines against malaria.FEMS Microbiol Rev. 2016 May;40(3):343-72. doi: 10.1093/femsre/fuw001. Epub 2016 Jan 31. FEMS Microbiol Rev. 2016. PMID: 26833236 Free PMC article. Review.
References
-
- Hall R, McBride J, Morgan G, Tait A, Zolg JW, et al. Antigens of the erythrocytes stages of the human malaria parasite Plasmodium falciparum detected by monoclonal antibodies. Mol Biochem Parasitol. 1983;7:247–265. - PubMed
-
- Gilson PR, O'Donnell RA, Nebl T, Sanders PR, Wickham ME, et al. MSP1(19) miniproteins can serve as targets for invasion inhibitory antibodies in Plasmodium falciparum provided they contain the correct domains for cell surface trafficking. Mol Microbiol. 2008;68:124–138. - PubMed
-
- Oka M, Aikawa M, Freeman RR, Holder AA, Fine E. Ultrastructural localization of protective antigens of Plasmodium yoelii merozoites by the use of monoclonal antibodies and ultrathin cryomicrotomy. Am J Trop Med Hyg. 1984;33:342–346. - PubMed
-
- Heidrich HG, Matzner M, Miettinen-Baumann A, Strych W. Immunoelectron microscopy shows that the 80,000-dalton antigen of Plasmodium falciparum merozoites is localized in the surface coat. Z Parasitenkd. 1986;72:681–683. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

