Cross-link formation of the cysteine 228-tyrosine 272 catalytic cofactor of galactose oxidase does not require dioxygen
- PMID: 18771294
- PMCID: PMC2762401
- DOI: 10.1021/bi8010835
Cross-link formation of the cysteine 228-tyrosine 272 catalytic cofactor of galactose oxidase does not require dioxygen
Abstract
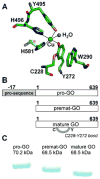
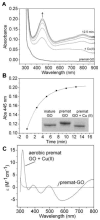
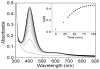
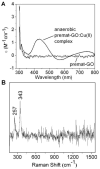
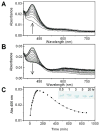
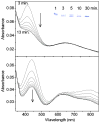
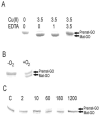
Galactose oxidase (GO) belongs to a class of proteins that self-catalyze assembly of their redox-active cofactors from active site amino acids. Generation of enzymatically active GO appears to require at least four sequential post-translational modifications: cleavage of a secretion signal sequence, copper-dependent cleavage of an N-terminal pro sequence, copper-dependent formation of a C228-Y272 thioether bond, and generation of the Y272 radical. The last two processes were investigated using a truncated protein (termed premat-GO) lacking the pro sequence and purified under copper-free conditions. Reactions of premat-GO with Cu(II) were investigated using optical, EPR, and resonance Raman spectroscopy, SDS-PAGE, and X-ray crystallography. Premat-GO reacted anaerobically with excess Cu(II) to efficiently form the thioether bond but not the Y272 radical. A potential C228-copper coordinated intermediate (lambda max = 406 nm) in the processing reaction, which had not yet formed the C228-Y272 cross-link, was identified from the absorption spectrum. A copper-thiolate protein complex, with copper coordinated to C228, H496, and H581, was also observed in a 3 min anaerobic soak by X-ray crystallography, whereas a 24 h soak revealed the C228-Y272 thioether bond. In solution, addition of oxygenated buffer to premat-GO preincubated with excess Cu(II) generated the Y272 radical state. On the basis of these data, a mechanism for the formation of the C228-Y272 bond and tyrosyl radical generation is proposed. The 406 nm complex is demonstrated to be a catalytically competent processing intermediate under anaerobic conditions. We propose a potential mechanism which is in common with aerobic processing by Cu(II) until the step at which the second electron acceptor is required.
Figures


Similar articles
-
Cu(I)-dependent biogenesis of the galactose oxidase redox cofactor.J Biol Chem. 2003 Jun 13;278(24):22090-101. doi: 10.1074/jbc.M300112200. Epub 2003 Apr 1. J Biol Chem. 2003. PMID: 12672814
-
Role of the Tyr-Cys cross-link to the active site properties of galactose oxidase.Inorg Chem. 2012 Mar 19;51(6):3513-24. doi: 10.1021/ic2022769. Epub 2012 Feb 28. Inorg Chem. 2012. PMID: 22372371
-
Formation of Monofluorinated Radical Cofactor in Galactose Oxidase through Copper-Mediated C-F Bond Scission.J Am Chem Soc. 2020 Nov 4;142(44):18753-18757. doi: 10.1021/jacs.0c08992. Epub 2020 Oct 22. J Am Chem Soc. 2020. PMID: 33091303 Free PMC article.
-
Cofactor processing in galactose oxidase.Biochem Soc Symp. 2004;(71):15-25. doi: 10.1042/bss0710015. Biochem Soc Symp. 2004. PMID: 15777009 Review.
-
Cofactor processing in galactose oxidase.Biochem Soc Trans. 2003 Jun;31(Pt 3):506-9. doi: 10.1042/bst10.1042/. Biochem Soc Trans. 2003. PMID: 12773145 Review.
Cited by
-
Proximity Labeling Facilitates Defining the Proteome Neighborhood of Photosystem II Oxygen Evolution Complex in a Model Cyanobacterium.Mol Cell Proteomics. 2022 Dec;21(12):100440. doi: 10.1016/j.mcpro.2022.100440. Epub 2022 Nov 8. Mol Cell Proteomics. 2022. PMID: 36356940 Free PMC article.
-
Copper active sites in biology.Chem Rev. 2014 Apr 9;114(7):3659-853. doi: 10.1021/cr400327t. Epub 2014 Mar 3. Chem Rev. 2014. PMID: 24588098 Free PMC article. Review. No abstract available.
-
Activation of dioxygen by copper metalloproteins and insights from model complexes.J Biol Inorg Chem. 2017 Apr;22(2-3):253-288. doi: 10.1007/s00775-016-1415-2. Epub 2016 Dec 5. J Biol Inorg Chem. 2017. PMID: 27921179 Free PMC article. Review.
-
Expression, purification, and characterization of galactose oxidase of Fusarium sambucinum in E. coli.Protein Expr Purif. 2015 Apr;108:73-79. doi: 10.1016/j.pep.2014.12.010. Epub 2014 Dec 24. Protein Expr Purif. 2015. PMID: 25543085 Free PMC article.
-
Spatially resolved proteomic mapping in living cells with the engineered peroxidase APEX2.Nat Protoc. 2016 Mar;11(3):456-75. doi: 10.1038/nprot.2016.018. Epub 2016 Feb 11. Nat Protoc. 2016. PMID: 26866790 Free PMC article.
References
-
- Okeley NM, van der Donk WA. Novel cofactors via post-translational modifications of enzyme active sites. Chem Biol. 2000;7:R159–R171. - PubMed
-
- Stubbe JA. Protein radical involvement in biological catalysis. Annu Rev Biochem. 1989;58:257–285. - PubMed
-
- Stubbe JA, van der Donk WA. Protein radicals in enzyme catalysis. Chem ReV. 1998;98:705–762. - PubMed
-
- Frey PA. Radical mechanisms of enzymatic catalysis. Annu Rev Biochem. 2001;70:121–148. - PubMed
-
- Banerjee R. Introduction: Radical enzymology. Chem Rev. 2003;103:2081–2081.
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources

