A divergent role for estrogen receptor-beta in node-positive and node-negative breast cancer classified according to molecular subtypes: an observational prospective study
- PMID: 18771580
- PMCID: PMC2614505
- DOI: 10.1186/bcr2139
A divergent role for estrogen receptor-beta in node-positive and node-negative breast cancer classified according to molecular subtypes: an observational prospective study
Abstract
Introduction: Estrogen receptor-alpha (ER-alpha) and progesterone receptor (PgR) are consolidated predictors of response to hormonal therapy (HT). In contrast, little information regarding the role of estrogen receptor-beta (ER-beta) in various breast cancer risk groups treated with different therapeutic regimens is available. In particular, there are no data concerning ER-beta distribution within the novel molecular breast cancer subtypes luminal A (LA) and luminal B (LB), HER2 (HS), and triple-negative (TN).
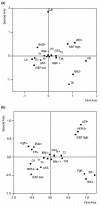
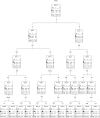
Methods: We conducted an observational prospective study using immunohistochemistry to evaluate ER-beta expression in 936 breast carcinomas. Associations with conventional biopathological factors and with molecular subtypes were analyzed by multiple correspondence analysis (MCA), while univariate and multivariate Cox regression analysis and classification and regression tree analysis were applied to determine the impact of ER-beta on disease-free survival in the 728 patients with complete follow-up data.
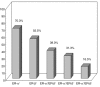
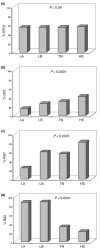
Results: ER-beta evenly distributes (55.5%) across the four molecular breast cancer subtypes, confirming the lack of correlation between ER-beta and classical prognosticators. However, the relationships among the biopathological factors, analyzed by MCA, showed that ER-beta positivity is located in the quadrant containing more aggressive phenotypes such as HER2 and TN or ER-alpha/PgR/Bcl2- tumors. Kaplan-Meier curves and Cox regression analysis identified ER-beta as a significant discriminating factor for disease-free survival both in the node-negative LA (P = 0.02) subgroup, where it is predictive of response to HT, and in the node-positive LB (P = 0.04) group, where, in association with PgR negativity, it conveys a higher risk of relapse.
Conclusion: Our data indicated that, in contrast to node-negative patients, in node-positive breast cancer patients, ER-beta positivity appears to be a biomarker related to a more aggressive clinical course. In this context, further investigations are necessary to better assess the role of the different ER-beta isoforms.
Figures


Comment in
-
The evolving role of oestrogen receptor beta in clinical breast cancer.Breast Cancer Res. 2008;10(5):111. doi: 10.1186/bcr2140. Epub 2008 Sep 19. Breast Cancer Res. 2008. PMID: 18831727 Free PMC article.
References
-
- Speirs V, Malone C, Walton DS, Kerin MJ, Atkin SL. Increased expression of estrogen receptor beta mRNA in tamoxifen-resistant breast cancer patients. Cancer Res. 1999;59:5421–5424. - PubMed
-
- Gruvberger-Saal SK, Bendahl PO, Saal LH, Laakso M, Hegardt C, Edén P, Peterson C, Malmström P, Isola J, Borg A, Fernö M. Estrogen receptor beta expression is associated with tamoxifen response in ERalpha-negative breast carcinoma. Clin Cancer Res. 2007;13:1987–1994. doi: 10.1158/1078-0432.CCR-06-1823. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

