CEP290 interacts with the centriolar satellite component PCM-1 and is required for Rab8 localization to the primary cilium
- PMID: 18772192
- PMCID: PMC2722899
- DOI: 10.1093/hmg/ddn277
CEP290 interacts with the centriolar satellite component PCM-1 and is required for Rab8 localization to the primary cilium
Abstract
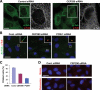
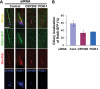
Joubert syndrome (JS) is a developmental brain disorder characterized by cerebellar vermis hypoplasia, abnormal eye movement, ataxia and mental retardation. Mutations in CEP290 mutations are responsible for the cerebello-oculo-renal subtype of JS that includes kidney cysts and retinal degeneration, two phenotypes commonly linked to ciliopathies. CEP290 mutations are also associated with Meckel-Gruber syndrome and Bardet-Biedl syndrome (BBS). Here we demonstrate that CEP290 interacts with a centriolar satellite protein PCM-1, which is implicated in BBS4 function. CEP290 binds to PCM-1 and localizes to centriolar satellites in a PCM-1- and microtubule-dependent manner. The depletion of CEP290 disrupts subcellular distribution and protein complex formation of PCM-1. In accord with PCM-1's role in microtubule organization, CEP290 knockdown causes the disorganization of the cytoplasmic microtubule network. Moreover, we show that both CEP290 and PCM-1 are required for ciliogenesis and are involved in the ciliary targeting of Rab8, a small GTPase shown to collaborate with BBS protein complex to promote ciliogenesis. Our results suggest that PCM-1 is a potential mediator that may link CEP290 with BBS proteins in common molecular pathways.
Figures



References
-
- Hildebrandt F., Otto E. Cilia and centrosomes: a unifying pathogenic concept for cystic kidney disease? Nat. Rev. Genet. 2005;6:928–940. - PubMed
-
- Badano J.L., Mitsuma N., Beales P.L., Katsanis N. The ciliopathies: an emerging class of human genetic disorders. Annu. Rev. Genomics Hum. Genet. 2006;7:125–148. - PubMed
-
- Valente E.M., Silhavy J.L., Brancati F., Barrano G., Krishnaswami S.R., Castori M., Lancaster M.A., Boltshauser E., Boccone L., Al-Gazali L., et al. Mutations in CEP290, which encodes a centrosomal protein, cause pleiotropic forms of Joubert syndrome. Nat. Genet. 2006;38:623–625. - PubMed
-
- Sayer J.A., Otto E.A., O’Toole J.F., Nurnberg G., Kennedy M.A., Becker C., Hennies H.C., Helou J., Attanasio M., Fausett B.V., et al. The centrosomal protein nephrocystin-6 is mutated in Joubert syndrome and activates transcription factor ATF4. Nat. Genet. 2006;38:674–681. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

