Yohimbine impairs extinction of cocaine-conditioned place preference in an alpha2-adrenergic receptor independent process
- PMID: 18772254
- PMCID: PMC2632785
- DOI: 10.1101/lm.1079308
Yohimbine impairs extinction of cocaine-conditioned place preference in an alpha2-adrenergic receptor independent process
Abstract
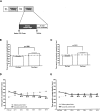
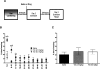
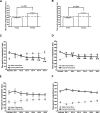
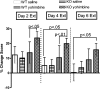
Extinction, a form of learning that has the ability to reshape learned behavior based on new experiences, has been heavily studied utilizing fear learning paradigms. Mechanisms underlying extinction of positive-valence associations, such as drug self-administration and place preference, are poorly understood yet may have important relevance to addiction treatment. Data suggest a major role for the noradrenergic system in extinction of fear-based learning. Employing both pharmacological and genetic approaches, we investigated the role of the alpha(2)-adrenergic receptor (alpha(2)-AR) in extinction of cocaine-conditioned place preference (CPP) and glutamatergic transmission in the bed nucleus of the stria terminalis (BNST). We found that pre-extinction systemic treatment with the alpha(2)-AR antagonist yohimbine impaired cocaine CPP extinction in C57BL/6J mice, an effect that was not mimicked by the more selective alpha(2)-AR antagonist, atipamezole. Moreover, alpha(2A)-AR knockout mice exhibited similar cocaine CPP extinction and exacerbated extinction impairing effects of yohimbine. Using acute brain slices and electrophysiological approaches, we found that yohimbine produces a slowly evolving depression of glutamatergic transmission in the BNST that was not mimicked by atipamezole. Further, this action was extant in slices from alpha(2A)-AR knockout mice. Our data strongly suggest that extinction-modifying effects of yohimbine are unlikely to be due to actions at alpha(2A)-ARs.
Figures

Similar articles
-
Pharmacological facilitation of fear extinction and the search for adjunct treatments for anxiety disorders--the case of yohimbine.Trends Pharmacol Sci. 2010 Jan;31(1):2-7. doi: 10.1016/j.tips.2009.10.003. Epub 2009 Dec 28. Trends Pharmacol Sci. 2010. PMID: 20036429 Free PMC article. Review.
-
Yohimbine depresses excitatory transmission in BNST and impairs extinction of cocaine place preference through orexin-dependent, norepinephrine-independent processes.Neuropsychopharmacology. 2012 Sep;37(10):2253-66. doi: 10.1038/npp.2012.76. Epub 2012 May 23. Neuropsychopharmacology. 2012. PMID: 22617356 Free PMC article.
-
Involvement of noradrenergic neurotransmission in the stress- but not cocaine-induced reinstatement of extinguished cocaine-induced conditioned place preference in mice: role for β-2 adrenergic receptors.Neuropsychopharmacology. 2010 Oct;35(11):2165-78. doi: 10.1038/npp.2010.86. Epub 2010 Jul 7. Neuropsychopharmacology. 2010. PMID: 20613718 Free PMC article.
-
A role for alpha-adrenergic receptors in extinction of conditioned fear and cocaine conditioned place preference.Behav Neurosci. 2010 Apr;124(2):204-10. doi: 10.1037/a0018909. Behav Neurosci. 2010. PMID: 20364880 Free PMC article.
-
A literature perspective on the pharmacological applications of yohimbine.Ann Med. 2022 Dec;54(1):2861-2875. doi: 10.1080/07853890.2022.2131330. Ann Med. 2022. PMID: 36263866 Free PMC article. Review.
Cited by
-
Enhancing Psychological Interventions for Post-Traumatic Stress Disorder (PTSD) Treatment with Memory Influencing Drugs.Curr Neuropharmacol. 2023;21(3):687-707. doi: 10.2174/1570159X21666221207162750. Curr Neuropharmacol. 2023. PMID: 36503450 Free PMC article. Review.
-
Pharmacological facilitation of fear extinction and the search for adjunct treatments for anxiety disorders--the case of yohimbine.Trends Pharmacol Sci. 2010 Jan;31(1):2-7. doi: 10.1016/j.tips.2009.10.003. Epub 2009 Dec 28. Trends Pharmacol Sci. 2010. PMID: 20036429 Free PMC article. Review.
-
Noradrenergic alpha-2 receptor modulators in the ventral bed nucleus of the stria terminalis: effects on anxiety behavior in postpartum and virgin female rats.Behav Neurosci. 2013 Aug;127(4):582-97. doi: 10.1037/a0032776. Epub 2013 Jun 24. Behav Neurosci. 2013. PMID: 23796237 Free PMC article.
-
Contrasting distribution of physiological cell types in different regions of the bed nucleus of the stria terminalis.J Neurophysiol. 2013 Nov;110(9):2037-49. doi: 10.1152/jn.00408.2013. Epub 2013 Aug 7. J Neurophysiol. 2013. PMID: 23926040 Free PMC article.
-
Repeated alcohol extinction sessions in conjunction with MK-801, but not yohimbine or propranolol, reduces subsequent alcohol cue-induced responding in rats.Pharmacol Biochem Behav. 2014 Jan;116:16-24. doi: 10.1016/j.pbb.2013.11.020. Epub 2013 Nov 22. Pharmacol Biochem Behav. 2014. PMID: 24269546 Free PMC article.
References
-
- Aghajanian G.K. Tolerance of locus coeruleus neurones to morphine and suppression of withdrawal response by clonidine. Nature. 1978;276:186–188. - PubMed
-
- Altman J.D., Trendelenburg A.U., MacMillan L., Bernstein D., Limbird L., Starke K., Kobilka B.K., Hein L. Abnormal regulation of the sympathetic nervous system in α2A-adrenergic receptor knockout mice. Mol. Pharmacol. 1999;56:154–161. - PubMed
-
- Aston-Jones G., Delfs J.M., Druhan J., Zhu Y. The bed nucleus of the stria terminalis. A target site for noradrenergic actions in opiate withdrawal. Ann. N. Y. Acad. Sci. 1999;877:486–498. - PubMed
-
- Bouton M.E. Context, ambiguity, and unlearning: Sources of relapse after behavioral extinction. Biol. Psychiatry. 2002;52:976–986. - PubMed
-
- Boyce-Rustay J.M., Palachick B., Hefner K., Chen Y.C., Karlsson R.M., Millstein R.A., Harvey-White J., Holmes A. Desipramine potentiation of the acute depressant effects of ethanol: Modulation by α2-adrenoreceptors and stress. Neuropharm. 2008 doi: 10.1016/j.neuropharm.2008.06.032. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
