Interaction between defects in ventilatory and thermoregulatory control in mice lacking 5-HT neurons
- PMID: 18775520
- PMCID: PMC2613046
- DOI: 10.1016/j.resp.2008.08.003
Interaction between defects in ventilatory and thermoregulatory control in mice lacking 5-HT neurons
Abstract
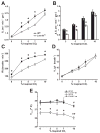
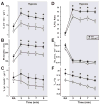
We have previously shown that mice with near-complete absence of 5-HT neurons (Lmx1bf/f/p) display a blunted hypercapnic ventilatory response (HCVR) and impaired cold-induced thermogenesis, but have normal baseline ventilation (), core body temperature (TCore) and hypoxic ventilatory responses (HVR) at warm ambient temperatures (TAmb; 30 degrees C). These results suggest that 5-HT neurons are an important site for integration of ventilatory, metabolic and temperature control. To better define this integrative role, we now determine how a moderate cold stress (TAmb of 25 degrees C) influences ventilatory control in adult Lmx1bf/f/p mice. During whole animal plethysmographic recordings at 25 degreesC, baseline , metabolic rate , and TCore of Lmx1bf/f/p mice were reduced (P < 0.001) compared to wild type (WT) mice. Additionally, the HCVR was reduced in Lmx1bf/f/p mice during normoxic (-33.1%) and hyperoxic (-40.9%) hypercapnia. However, in Lmx1bf/f/p mice was equal to that in WT mice while breathing 10% CO2, indicating that non-5-HT neurons may play a dominant role during extreme hypercapnia. Additionally, ventilation was decreased during hypoxia in Lmx1bf/f/p mice compared to WT mice at 25 degrees C due to decreased TCore. These data suggest that a moderate cold stress in Lmx1bf/f/p mice leads to further dysfunction in ventilatory control resulting from failure to adequately maintain TCore. We conclude that 5-HT neurons contribute to the hypercapnic ventilatory response under physiologic, more than during extreme levels of CO2, and that mild cold stress further compromises ventilatory control in Lmx1bf/f/p mice as a result of defective thermogenesis.
Figures


Similar articles
-
Medullary 5-HT neurons: Switch from tonic respiratory drive to chemoreception during postnatal development.Neuroscience. 2017 Mar 6;344:1-14. doi: 10.1016/j.neuroscience.2016.09.002. Epub 2016 Sep 9. Neuroscience. 2017. PMID: 27619736 Free PMC article.
-
Defects in breathing and thermoregulation in mice with near-complete absence of central serotonin neurons.J Neurosci. 2008 Mar 5;28(10):2495-505. doi: 10.1523/JNEUROSCI.4729-07.2008. J Neurosci. 2008. PMID: 18322094 Free PMC article.
-
Altered ventilatory and thermoregulatory control in male and female adult Pet-1 null mice.Respir Physiol Neurobiol. 2011 Jul 31;177(2):133-40. doi: 10.1016/j.resp.2011.03.020. Epub 2011 Mar 29. Respir Physiol Neurobiol. 2011. PMID: 21453797 Free PMC article.
-
Brainstem serotonin deficiency in the neonatal period: autonomic dysregulation during mild cold stress.J Physiol. 2011 Apr 15;589(Pt 8):2055-64. doi: 10.1113/jphysiol.2010.203679. Epub 2011 Feb 28. J Physiol. 2011. PMID: 21486799 Free PMC article.
-
The role of medullary serotonin (5-HT) neurons in respiratory control: contributions to eupneic ventilation, CO2 chemoreception, and thermoregulation.J Appl Physiol (1985). 2010 May;108(5):1425-32. doi: 10.1152/japplphysiol.01270.2009. Epub 2010 Feb 4. J Appl Physiol (1985). 2010. PMID: 20133432 Free PMC article. Review.
Cited by
-
Effects of dorsomedial medullary 5-HT2 receptor antagonism on initial ventilatory airway responses to hypercapnic hypoxia in mice.Exp Brain Res. 2013 Oct;230(4):547-54. doi: 10.1007/s00221-013-3493-5. Epub 2013 Mar 30. Exp Brain Res. 2013. PMID: 23543107
-
Methods for the Comprehensive in vivo Analysis of Energy Flux, Fluid Homeostasis, Blood Pressure, and Ventilatory Function in Rodents.Front Physiol. 2022 Feb 25;13:855054. doi: 10.3389/fphys.2022.855054. eCollection 2022. Front Physiol. 2022. PMID: 35283781 Free PMC article.
-
Chronic central serotonin depletion attenuates ventilation and body temperature in young but not adult Tph2 knockout rats.J Appl Physiol (1985). 2016 May 1;120(9):1070-81. doi: 10.1152/japplphysiol.01015.2015. Epub 2016 Feb 11. J Appl Physiol (1985). 2016. PMID: 26869713 Free PMC article.
-
The sleep-wake cycle and motor activity, but not temperature, are disrupted over the light-dark cycle in mice genetically depleted of serotonin.Am J Physiol Regul Integr Comp Physiol. 2015 Jan 1;308(1):R10-7. doi: 10.1152/ajpregu.00400.2014. Epub 2014 Nov 12. Am J Physiol Regul Integr Comp Physiol. 2015. PMID: 25394829 Free PMC article.
-
Medullary 5-HT neurons: Switch from tonic respiratory drive to chemoreception during postnatal development.Neuroscience. 2017 Mar 6;344:1-14. doi: 10.1016/j.neuroscience.2016.09.002. Epub 2016 Sep 9. Neuroscience. 2017. PMID: 27619736 Free PMC article.
References
-
- Al-Zubaidy ZA, Erickson RL, Greer JJ. Serotonergic and noradrenergic effects on respiratory neural discharge in the medullary slice preparation of neonatal rats. Pflugers Arch. 1996;431:942–949. - PubMed
-
- Bradley SR, Pieribone VA, Wang W, Severson CA, Jacobs RA, Richerson GB. Chemosensitive serotonergic neurons are closely associated with large medullary arteries. Nat Neurosci. 2002;5:401–402. - PubMed
-
- Cinelli P, Rettich A, Seifert B, Burki K, Arras M. Comparative analysis and physiological impact of different tissue biopsy methodologies used for the genotyping of laboratory mice. Lab Anim. 2007;41:174–184. - PubMed
-
- Deng BS, Nakamura A, Zhang W, Yanagisawa M, Fukuda Y, Kuwaki T. Contribution of orexin in hypercapnic chemoreflex: Evidence from genetic and pharmacological disruption and supplementation studies in mice. J Appl Physiol 2007 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

