Interferon regulatory factor 4 and 8 in B-cell development
- PMID: 18775669
- PMCID: PMC2823592
- DOI: 10.1016/j.it.2008.07.006
Interferon regulatory factor 4 and 8 in B-cell development
Abstract
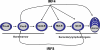
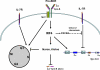
Interferon regulatory factor 4 (IRF4) and 8 are members of the interferon regulatory factor family of transcription factors and have been shown to be essential for the development and function of T cells, macrophages and dendritic cells. A series of recent studies have further demonstrated critical functions for IRF4 and 8 at several stages of B-cell development including pre-B-cell development, receptor editing, germinal center reaction and plasma cell generation. Collectively, these new studies provide molecular insights into the function of IRF4 and 8 and underscore a requirement for IRF4 and 8 throughout B-cell development. This review focuses on the recent advances on the roles of IRF4 and 8 in B-cell development.
Figures
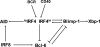
References
-
- Tamura T, Yanai H, Savitsky D, Taniguchi T. The IRF family transcription factors in immunity and oncogenesis. Annu Rev Immunol. 2008;26:535–584. - PubMed
-
- Eisenbeis CF, Singh H, Storb U. Pip, a novel IRF family member, is a lymphoid-specific, PU.1-dependent transcriptional activator. Genes Dev. 1995;9:1377–1387. - PubMed
-
- Brass AL, Kehrli E, Eisenbeis CF, Storb U, Singh H. Pip, a lymphoid-restricted IRF, contains a regulatory domain that is important for autoinhibition and ternary complex formation with the Ets factor PU.1. Genes Dev. 1996;10:2335–2347. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

