ThPOK acts late in specification of the helper T cell lineage and suppresses Runx-mediated commitment to the cytotoxic T cell lineage
- PMID: 18776905
- PMCID: PMC2666788
- DOI: 10.1038/ni.1652
ThPOK acts late in specification of the helper T cell lineage and suppresses Runx-mediated commitment to the cytotoxic T cell lineage
Abstract
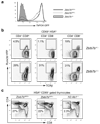
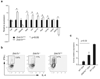
The transcription factor ThPOK is required and sufficient for the generation of CD4(+)CD8(-) thymocytes, yet the mechanism by which ThPOK orchestrates differentiation into the CD4(+) helper T cell lineage remains unclear. Here we used reporter mice to track the expression of transcription factors in developing thymocytes. Distal promoter-driven expression of the gene encoding the transcription factor Runx3 was restricted to major histocompatibility complex (MHC) class I-selected thymocytes. In ThPOK-deficient mice, such expression was derepressed in MHC class II-selected thymocytes, which contributed to their redirection to the CD8(+) T cell lineage. In the absence of both ThPOK and Runx, redirection was prevented and cells potentially belonging to the CD4(+) lineage, presumably specified independently of ThPOK, were generated. Our results suggest that MHC class II-selected thymocytes are directed toward the CD4(+) lineage independently of ThPOK but require ThPOK to prevent Runx-dependent differentiation toward the CD8(+) lineage.
Figures




Comment in
-
Decision by committee: new light on the CD4/CD8-lineage choice.Immunol Cell Biol. 2009 Feb;87(2):109-12. doi: 10.1038/icb.2008.100. Epub 2008 Dec 16. Immunol Cell Biol. 2009. PMID: 19079359 No abstract available.
References
-
- Kappes DJ, He X, He X. CD4-CD8 lineage commitment: an inside view. Nat Immunol. 2005;6:761–766. - PubMed
-
- Zou YR, et al. Epigenetic silencing of CD4 in T cells committed to the cytotoxic lineage. Nat Genet. 2001;29:332–336. - PubMed
-
- Sawada S, Scarborough JD, Killeen N, Littman DR. A lineage-specific transcriptional silencer regulates CD4 gene expression during T lymphocyte development. Cell. 1994;77:917–929. - PubMed
-
- Kioussis D, Ellmeier W. Chromatin and CD4, CD8A and CD8B gene expression during thymic differentiation. Nat Rev Immunol. 2002;2:909–919. - PubMed
-
- Laslo P, et al. Multilineage transcriptional priming and determination of alternate hematopoietic cell fates. Cell. 2006;126:755–766. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

