Distinct tumor suppressor mechanisms evolve in rodent species that differ in size and lifespan
- PMID: 18778411
- PMCID: PMC2637185
- DOI: 10.1111/j.1474-9726.2008.00431.x
Distinct tumor suppressor mechanisms evolve in rodent species that differ in size and lifespan
Abstract
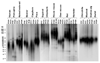
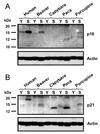
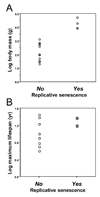
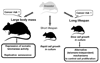
Large, long-lived species experience more lifetime cell divisions and hence a greater risk of spontaneous tumor formation than smaller, short-lived species. Large, long-lived species are thus expected to evolve more elaborate tumor suppressor systems. In previous work, we showed that telomerase activity coevolves with body mass, but not lifespan, in rodents: telomerase activity is repressed in the somatic tissues of large rodent species but remains active in small ones. Without telomerase activity, the telomeres of replicating cells become progressively shorter until, at some critical length, cells stop dividing. Our findings therefore suggested that repression of telomerase activity mitigates the increased risk of cancer in larger-bodied species but not necessarily longer-lived ones. These findings imply that other tumor suppressor mechanisms must mitigate increased cancer risk in long-lived species. Here, we examined the proliferation of fibroblasts from 15 rodent species with diverse body sizes and lifespans. We show that, consistent with repressed telomerase activity, fibroblasts from large rodents undergo replicative senescence accompanied by telomere shortening and overexpression of p16(Ink4a) and p21(Cip1/Waf1) cycline-dependent kinase inhibitors. Interestingly, small rodents with different lifespans show a striking difference: cells from small shorter-lived species display continuous rapid proliferation, whereas cells from small long-lived species display continuous slow proliferation. We hypothesize that cells of small long-lived rodents, lacking replicative senescence, have evolved alternative tumor-suppressor mechanisms that prevent inappropriate cell division in vivo and slow cell growth in vitro. Thus, large-bodied species and small but long-lived species have evolved distinct tumor suppressor mechanisms.
Figures




Similar articles
-
Telomerase activity coevolves with body mass not lifespan.Aging Cell. 2007 Feb;6(1):45-52. doi: 10.1111/j.1474-9726.2006.00262.x. Epub 2006 Dec 14. Aging Cell. 2007. PMID: 17173545 Free PMC article.
-
Evolution of telomere maintenance and tumour suppressor mechanisms across mammals.Philos Trans R Soc Lond B Biol Sci. 2018 Mar 5;373(1741):20160443. doi: 10.1098/rstb.2016.0443. Philos Trans R Soc Lond B Biol Sci. 2018. PMID: 29335367 Free PMC article.
-
Comparative biology of mammalian telomeres: hypotheses on ancestral states and the roles of telomeres in longevity determination.Aging Cell. 2011 Oct;10(5):761-8. doi: 10.1111/j.1474-9726.2011.00718.x. Epub 2011 Jun 1. Aging Cell. 2011. PMID: 21518243 Free PMC article.
-
Coevolution of telomerase activity and body mass in mammals: from mice to beavers.Mech Ageing Dev. 2009 Jan-Feb;130(1-2):3-9. doi: 10.1016/j.mad.2008.02.008. Epub 2008 Feb 23. Mech Ageing Dev. 2009. PMID: 18387652 Free PMC article. Review.
-
Telomere length maintenance in aging and carcinogenesis.Int J Oncol. 2000 Nov;17(5):981-9. doi: 10.3892/ijo.17.5.981. Int J Oncol. 2000. PMID: 11029502 Review.
Cited by
-
Quantitative model of cell cycle arrest and cellular senescence in primary human fibroblasts.PLoS One. 2012;7(8):e42150. doi: 10.1371/journal.pone.0042150. Epub 2012 Aug 7. PLoS One. 2012. PMID: 22879912 Free PMC article.
-
The three dimensions of somatic evolution: Integrating the role of genetic damage, life-history traits, and aging in carcinogenesis.Evol Appl. 2020 Mar 20;13(7):1569-1580. doi: 10.1111/eva.12947. eCollection 2020 Aug. Evol Appl. 2020. PMID: 32821273 Free PMC article.
-
Can Peto's paradox be used as the null hypothesis to identify the role of evolution in natural resistance to cancer? A critical review.BMC Cancer. 2015 Oct 24;15:792. doi: 10.1186/s12885-015-1782-z. BMC Cancer. 2015. PMID: 26499116 Free PMC article. Review.
-
Senescence in the aging process.F1000Res. 2017 Jul 25;6:1219. doi: 10.12688/f1000research.10903.1. eCollection 2017. F1000Res. 2017. PMID: 28781767 Free PMC article. Review.
-
Selective anticancer agents suppress aging in Drosophila.Oncotarget. 2013 Sep;4(9):1507-26. doi: 10.18632/oncotarget.1272. Oncotarget. 2013. PMID: 24096697 Free PMC article.
References
-
- Adkins RM, Walton AH, Honeycutt RL. Higher-level systematics of rodents and divergence time estimates based on two congruent nuclear genes. Mol Phylogenet Evol. 2003;26:409–420. - PubMed
-
- Argyle D, Ellsmore V, Gault EA, Munro AF, Nasir L. Equine telomeres and telomerase in cellular immortalisation and ageing. Mech Ageing Dev. 2003;124:759–764. - PubMed
-
- Austad SN. Diverse aging rates in metazoans: targets for functional genomics. Mech Ageing Dev. 2005;126:43–49. - PubMed
-
- Austad SN, Fischer KE. Mammalian aging, metabolism, and ecology: evidence from the bats and marsupials. J Gerontol. 1991;46:B47–B53. - PubMed
-
- Bekaert S, Derradji H, Baatout S. Telomere biology in mammalian germ cells and during development. Dev Biol. 2004;274:15–30. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

