STAT92E is a positive regulator of Drosophila inhibitor of apoptosis 1 (DIAP/1) and protects against radiation-induced apoptosis
- PMID: 18779571
- PMCID: PMC2544535
- DOI: 10.1073/pnas.0806291105
STAT92E is a positive regulator of Drosophila inhibitor of apoptosis 1 (DIAP/1) and protects against radiation-induced apoptosis
Abstract
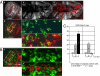
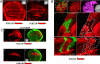
The proapoptotic factors Reaper, Hid, Grim, and Sickle regulate apoptosis in Drosophila by inhibiting the antiapoptotic factor DIAP1 (Drosophila inhibitor of apoptosis 1). Heat, UV light, x-rays, and developmental signals can all increase the proapoptotic factors, but the control of transcription of the diap1 gene is unclear. We show that in imaginal discs the single Drosophila STAT protein (STAT92E) when activated can directly increase DIAP1 through binding to STAT DNA-binding sites in the diap1 promoter. The STAT92E contribution to DIAP1 production is required for cell survival after x-irradiation but not under unstressed conditions. Because DIAP1 prevents apoptosis after a variety of stresses, STAT92E may have a role in regulating stress responses in general.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Wahl GM, Carr AM. The evolution of diverse biological responses to DNA damage: Insights from yeast and p53. Nat Cell Biol. 2001;3:E277–E286. - PubMed
-
- Kornbluth S, White K. Apoptosis in Drosophila: Neither fish nor fowl (nor man, nor worm) J Cell Sci. 2005;118:1779–1787. - PubMed
-
- Wang SL, Hawkins CJ, Yoo SJ, Müller HA, Hay BA. The Drosophila caspase inhibitor DIAP1 is essential for cell survival and is negatively regulated by HID. Cell. 1999;98:453–463. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

