Terbutaline and the prevention of nocturnal hypoglycemia in type 1 diabetes
- PMID: 18782903
- PMCID: PMC2584177
- DOI: 10.2337/dc08-0520
Terbutaline and the prevention of nocturnal hypoglycemia in type 1 diabetes
Abstract
Objective: Bedtime administration of 5.0 mg of the beta(2)-adrenergic agonist terbutaline prevents nocturnal hypoglycemia but causes morning hyperglycemia in type 1 diabetes. We tested the hypothesis that 2.5 mg terbutaline prevents nocturnal hypoglycemia without causing morning hyperglycemia.
Research design and methods: This was a randomized double-blind crossover pilot study (placebo, 2.5 mg terbutaline, and 5.0 mg terbutaline) in 15 patients with type 1 diabetes.
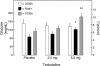
Results: Mean +/- SE nadir nocturnal plasma glucose concentrations were 87 +/- 14 mg/dl following placebo, 100 +/- 14 mg/dl following 2.5 mg terbutaline, and 122 +/- 13 mg/dl following 5.0 mg terbutaline (P < 0.05 vs. placebo). Nadir levels were <50 mg/dl in 5, 2, and 0 patients (P < 0.05 vs. placebo), respectively. Morning levels were 113 +/- 18, 127 +/- 17, and 183 +/- 19 mg/dl (P < 0.02 vs. placebo), respectively.
Conclusions: Terbutaline may be shown to be effective and safe in the prevention of nocturnal hypoglycemia in type 1 diabetes in a suitably powered randomized controlled trial.
Figures
Similar articles
-
Preventing post-exercise nocturnal hypoglycemia in children with type 1 diabetes.J Pediatr. 2010 Nov;157(5):784-8.e1. doi: 10.1016/j.jpeds.2010.06.004. Epub 2010 Jul 21. J Pediatr. 2010. PMID: 20650471 Free PMC article. Clinical Trial.
-
Alanine and terbutaline in the prevention of nocturnal hypoglycemia in IDDM.Diabetes Care. 1997 Aug;20(8):1231-6. doi: 10.2337/diacare.20.8.1231. Diabetes Care. 1997. PMID: 9250445 Clinical Trial.
-
Nocturnal hypoglycemia in type 1 diabetes: an assessment of preventive bedtime treatments.J Clin Endocrinol Metab. 2006 Jun;91(6):2087-92. doi: 10.1210/jc.2005-2798. Epub 2006 Feb 21. J Clin Endocrinol Metab. 2006. PMID: 16492699 Clinical Trial.
-
Nocturnal hypoglycemia: clinical manifestations and therapeutic strategies toward prevention.Endocr Pract. 2003 Nov-Dec;9(6):530-43. doi: 10.4158/EP.9.6.530. Endocr Pract. 2003. PMID: 14715482 Review.
-
The efficacy and safety of insulin pump therapy with predictive low glucose suspend feature in decreasing hypoglycemia in children with type 1 diabetes mellitus: A systematic review and meta-analysis.Pediatr Diabetes. 2020 Nov;21(7):1256-1267. doi: 10.1111/pedi.13088. Epub 2020 Sep 14. Pediatr Diabetes. 2020. PMID: 32738022
Cited by
-
β2-Adrenergic receptor agonist administration promotes counter-regulatory responses and recovery from hypoglycaemia in rats.Diabetologia. 2013 Nov;56(11):2517-23. doi: 10.1007/s00125-013-3009-7. Epub 2013 Aug 10. Diabetologia. 2013. PMID: 23933834 Free PMC article.
-
Hypoglycemia: from the laboratory to the clinic.Diabetes Care. 2009 Aug;32(8):1364-71. doi: 10.2337/dc09-0113. Diabetes Care. 2009. PMID: 19638523 Free PMC article. No abstract available.
-
Potential approaches to prevent hypoglycemia-associated autonomic failure.J Investig Med. 2018 Mar;66(3):641-647. doi: 10.1136/jim-2017-000582. Epub 2017 Nov 14. J Investig Med. 2018. PMID: 29141871 Free PMC article. Review.
-
Preventing post-exercise nocturnal hypoglycemia in children with type 1 diabetes.J Pediatr. 2010 Nov;157(5):784-8.e1. doi: 10.1016/j.jpeds.2010.06.004. Epub 2010 Jul 21. J Pediatr. 2010. PMID: 20650471 Free PMC article. Clinical Trial.
-
The effect of terbutaline on the absorption of pulmonary administered insulin in subjects with asthma.Br J Clin Pharmacol. 2010 Mar;69(3):271-8. doi: 10.1111/j.1365-2125.2009.03573.x. Br J Clin Pharmacol. 2010. PMID: 20233198 Free PMC article. Clinical Trial.
References
-
- Cryer PE: Diverse causes of hypoglycemia-associated autonomic failure in diabetes. N Engl J Med 350:2272–2279, 2004 - PubMed
-
- The Diabetes Control and Complications Trial Research Group: Hypoglycemia in the Diabetes Control and Complications Trial. Diabetes 46:271–286, 1997 - PubMed
-
- Raju B, Arbelaez AM, Breckenridge SM, Cryer PE: Nocturnal hypoglycemia in type 1 diabetes: an assessment of preventive bedtime treatments. J Clin Endocrinol Metab 91:2087–2092, 2006 - PubMed
-
- Wentholt IME, Maran A, Masurel N, Heine RJ, Hoekstra JBL, DeVries JH: Nocturnal hypoglycaemia in type 1 diabetic patients, assessed with continuous glucose monitoring: frequency, duration and associations. Diabet Med 24:527–532, 2007 - PubMed
-
- Jones TW, Porter P, Sherwin RS, Davis EA, O'Leary P, Frazer F, Byrne G, Stick S, Tamborlane WV: Decreased epinephrine responses to hypoglycemia during sleep. N Engl J Med 338:1657–1662, 1998 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical