Expression of nonstructural rotavirus protein NSP4 mimics Ca2+ homeostasis changes induced by rotavirus infection in cultured cells
- PMID: 18787006
- PMCID: PMC2573286
- DOI: 10.1128/JVI.00577-08
Expression of nonstructural rotavirus protein NSP4 mimics Ca2+ homeostasis changes induced by rotavirus infection in cultured cells
Abstract
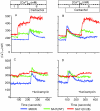
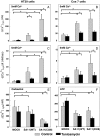
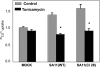
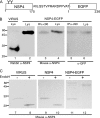
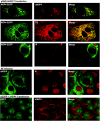
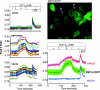
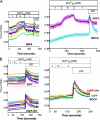
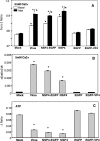
Rotavirus infection modifies Ca(2+) homeostasis, provoking an increase in Ca(2+) permeation, the cytoplasmic Ca(2+) concentration ([Ca(2+)](cyto)), and total Ca(2+) pools and a decrease in Ca(2+) response to agonists. A glycosylated viral protein(s), NSP4 and/or VP7, may be responsible for these effects. HT29 or Cos-7 cells were infected by the SA11 clone 28 strain, in which VP7 is not glycosylated, or transiently transfected with plasmids coding for NSP4-enhanced green fluorescent protein (EGFP) or NSP4. The permeability of the plasma membrane to Ca(2+) and the amount of Ca(2+) sequestered in the endoplasmic reticulum released by carbachol or ATP were measured in fura-2-loaded cells at the single-cell level under a fluorescence microscope or in cell suspensions in a fluorimeter. Total cell Ca(2+) pools were evaluated as (45)Ca(2+) uptake. Infection with SA11 clone 28 induced an increase in Ca(2+) permeability and (45)Ca(2+) uptake similar to that found with the normally glycosylated SA11 strain. These effects were inhibited by tunicamycin, indicating that inhibition of glycosylation of a viral protein other than VP7 affects the changes of Ca(2+) homeostasis induced by infection. Expression of NSP4-EGFP or NSP4 in transfected cells induced the same changes observed with rotavirus infection, whereas the expression of EGFP or EGFP-VP4 showed the behavior of uninfected and untransfected cells. Increased (45)Ca(2+) uptake was also observed in cells expressing NSP4-EGFP or NSP4, as evidenced in rotavirus infection. These results indicate that glycosylated NSP4 is primarily responsible for altering the Ca(2+) homeostasis of infected cells through an initial increase of cell membrane permeability to Ca(2+).
Figures

Similar articles
-
Silencing of rotavirus NSP4 or VP7 expression reduces alterations in Ca2+ homeostasis induced by infection of cultured cells.J Virol. 2008 Jun;82(12):5815-24. doi: 10.1128/JVI.02719-07. Epub 2008 Apr 9. J Virol. 2008. PMID: 18400845 Free PMC article.
-
Dissecting the Ca²⁺ entry pathways induced by rotavirus infection and NSP4-EGFP expression in Cos-7 cells.Virus Res. 2012 Aug;167(2):285-96. doi: 10.1016/j.virusres.2012.05.012. Epub 2012 May 23. Virus Res. 2012. PMID: 22634036
-
The rotavirus nonstructural glycoprotein NSP4 mobilizes Ca2+ from the endoplasmic reticulum.J Virol. 1995 Sep;69(9):5763-72. doi: 10.1128/JVI.69.9.5763-5772.1995. J Virol. 1995. PMID: 7637021 Free PMC article.
-
Rotavirus protein expression is important for virus assembly and pathogenesis.Arch Virol Suppl. 1996;12:69-77. doi: 10.1007/978-3-7091-6553-9_8. Arch Virol Suppl. 1996. PMID: 9015103 Review.
-
Rotavirus NSP4: a multifunctional viral enterotoxin.Viral Immunol. 2005;18(1):27-40. doi: 10.1089/vim.2005.18.27. Viral Immunol. 2005. PMID: 15802952 Review. No abstract available.
Cited by
-
Toxin mediated diarrhea in the 21 century: the pathophysiology of intestinal ion transport in the course of ETEC, V. cholerae and rotavirus infection.Toxins (Basel). 2010 Aug;2(8):2132-57. doi: 10.3390/toxins2082132. Epub 2010 Aug 10. Toxins (Basel). 2010. PMID: 22069677 Free PMC article. Review.
-
Isolation and Pathogenicity Analysis of a G5P[23] Porcine Rotavirus Strain.Viruses. 2023 Dec 22;16(1):21. doi: 10.3390/v16010021. Viruses. 2023. PMID: 38257722 Free PMC article.
-
Rotavirus Calcium Dysregulation Manifests as Dynamic Calcium Signaling in the Cytoplasm and Endoplasmic Reticulum.Sci Rep. 2019 Jul 25;9(1):10822. doi: 10.1038/s41598-019-46856-8. Sci Rep. 2019. PMID: 31346185 Free PMC article.
-
Rotavirus infection activates the UPR but modulates its activity.Virol J. 2011 Jul 20;8:359. doi: 10.1186/1743-422X-8-359. Virol J. 2011. PMID: 21774819 Free PMC article.
-
Virus Control of Cell Metabolism for Replication and Evasion of Host Immune Responses.Front Cell Infect Microbiol. 2019 Apr 18;9:95. doi: 10.3389/fcimb.2019.00095. eCollection 2019. Front Cell Infect Microbiol. 2019. PMID: 31058096 Free PMC article. Review.
References
-
- Altenburg, B. C., D. Y. Graham, and M. K. Estes. 1980. Ultrastructural study of rotavirus replication in cultured cells. J. Gen. Virol. 4675-85. - PubMed
-
- Ball, J. M., P. Tian, C. Q. Zeng, A. P. Morris, and M. K. Estes. 1996. Age-dependent diarrhea induced by a rotaviral nonstructural glycoprotein. Science 272101-104. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

