Role of dosage-sensitive sex reversal, adrenal hypoplasia congenita, critical region on the X chromosome, gene 1 in protein kinase A- and protein kinase C-mediated regulation of the steroidogenic acute regulatory protein expression in mouse Leydig tumor cells: mechanism of action
- PMID: 18787026
- PMCID: PMC2630909
- DOI: 10.1210/en.2008-0368
Role of dosage-sensitive sex reversal, adrenal hypoplasia congenita, critical region on the X chromosome, gene 1 in protein kinase A- and protein kinase C-mediated regulation of the steroidogenic acute regulatory protein expression in mouse Leydig tumor cells: mechanism of action
Abstract
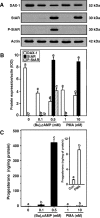
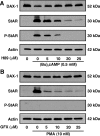
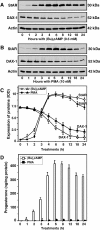
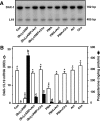
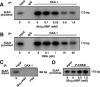
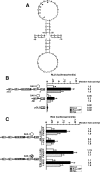
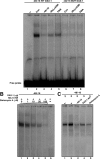
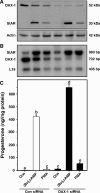
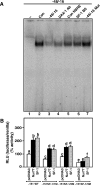
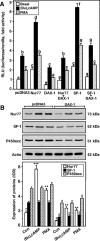
Dosage-sensitive sex reversal, adrenal hypoplasia congenita, critical region on the X chromosome, gene 1 (DAX-1) is an orphan nuclear receptor that has been demonstrated to be instrumental to the expression of the steroidogenic acute regulatory (StAR) protein that regulates steroid biosynthesis in steroidogenic cells. However, its mechanism of action remains obscure. The present investigation was aimed at exploring the molecular involvement of DAX-1 in protein kinase A (PKA)- and protein kinase C (PKC)-mediated regulation of StAR expression and its concomitant impact on steroid synthesis using MA-10 mouse Leydig tumor cells. We demonstrate that activation of the PKA and PKC pathways, by a cAMP analog dibutyryl (Bu)2cAMP [(Bu)2cAMP] and phorbol 12-myristate 13-acetate (PMA), respectively, markedly decreased DAX-1 expression, an event that was inversely correlated with StAR protein, StAR mRNA, and progesterone levels. Notably, the suppression of DAX-1 requires de novo transcription and translation, suggesting that the effect of DAX-1 in regulating StAR expression is dynamic. Chromatin immunoprecipitation studies revealed the association of DAX-1 with the proximal but not the distal region of the StAR promoter, and both (Bu)2cAMP and PMA decreased in vivo DAX-1-DNA interactions. EMSA and reporter gene analyses demonstrated the functional integrity of this interaction by showing that DAX-1 binds to a DNA hairpin at position -44/-20 bp of the mouse StAR promoter and that the binding of DAX-1 to this region decreases progesterone synthesis by impairing transcription of the StAR gene. In support of this, targeted silencing of endogenous DAX-1 elevated basal, (Bu)2cAMP-, and PMA-stimulated StAR expression and progesterone synthesis. Transrepression of the StAR gene by DAX-1 was tightly associated with expression of the nuclear receptors Nur77 and steroidogenic factor-1, demonstrating these factors negatively modulate the steroidogenic response. These findings provide insight into the molecular events by which DAX-1 influences the PKA and PKC signaling pathways involved in the regulation of the StAR protein and steroidogenesis in mouse Leydig tumor cells.
Figures
Similar articles
-
Regulation of Leydig cell steroidogenesis by extracellular signal-regulated kinase 1/2: role of protein kinase A and protein kinase C signaling.J Endocrinol. 2007 Apr;193(1):53-63. doi: 10.1677/JOE-06-0201. J Endocrinol. 2007. PMID: 17400803
-
Involvement of protein kinase C and cyclic adenosine 3',5'-monophosphate-dependent kinase in steroidogenic acute regulatory protein expression and steroid biosynthesis in Leydig cells.Biol Reprod. 2005 Aug;73(2):244-55. doi: 10.1095/biolreprod.104.037721. Epub 2005 Apr 6. Biol Reprod. 2005. PMID: 15814901
-
Regulation of steroidogenesis and steroidogenic acute regulatory protein in R2C cells by DAX-1 (dosage-sensitive sex reversal, adrenal hypoplasia congenita, critical region on the X chromosome, gene-1).Endocrinology. 2004 Dec;145(12):5629-37. doi: 10.1210/en.2004-0941. Epub 2004 Sep 9. Endocrinology. 2004. PMID: 15358680
-
DAX-1, an 'antitestis' gene.Cell Mol Life Sci. 1999 Jun;55(6-7):857-63. doi: 10.1007/pl00013201. Cell Mol Life Sci. 1999. PMID: 10412368 Free PMC article. Review.
-
The gene responsible for adrenal hypoplasia congenita, DAX-1, encodes a nuclear hormone receptor that defines a new class within the superfamily.Recent Prog Horm Res. 1996;51:241-59; discussion 259-60. Recent Prog Horm Res. 1996. PMID: 8701082 Review.
Cited by
-
Retinoid regulated macrophage cholesterol efflux involves the steroidogenic acute regulatory protein.Data Brief. 2016 Mar 19;7:940-5. doi: 10.1016/j.dib.2016.03.055. eCollection 2016 Jun. Data Brief. 2016. PMID: 27081671 Free PMC article.
-
Current knowledge on the acute regulation of steroidogenesis.Biol Reprod. 2018 Jul 1;99(1):13-26. doi: 10.1093/biolre/ioy102. Biol Reprod. 2018. PMID: 29718098 Free PMC article. Review.
-
Extra-adrenal glucocorticoid biosynthesis: implications for autoimmune and inflammatory disorders.Genes Immun. 2020 May;21(3):150-168. doi: 10.1038/s41435-020-0096-6. Epub 2020 Mar 23. Genes Immun. 2020. PMID: 32203088 Free PMC article. Review.
-
The Effect of Cordycepin on Steroidogenesis and Apoptosis in MA-10 Mouse Leydig Tumor Cells.Evid Based Complement Alternat Med. 2011;2011:750468. doi: 10.1155/2011/750468. Epub 2011 Jun 5. Evid Based Complement Alternat Med. 2011. PMID: 21716681 Free PMC article.
-
Role of basic leucine zipper proteins in transcriptional regulation of the steroidogenic acute regulatory protein gene.Mol Cell Endocrinol. 2009 Apr 10;302(1):1-11. doi: 10.1016/j.mce.2008.12.009. Epub 2008 Dec 25. Mol Cell Endocrinol. 2009. PMID: 19150388 Free PMC article. Review.
References
-
- Zanaria E, Muscatelli F, Bardoni B, Strom TM, Guioli S, Guo W, Lalli E, Moser C, Walker AP, McCabe ER, Meitinger T, Monaco AP, Sassone-Corsi P, Camerino G An unusual member of the nuclear hormone receptor superfamily responsible for X-linked adrenal hypoplasia congenita. Nature 372:635–641 - PubMed
-
- Muscatelli F, Strom TM, Walker AP, Zanaria E, Recan D, Meindl A, Bardoni B, Guioli S, Zehetner G, Rabl W, Schwarz HP, Kaplan J-C, Camerino G, Meitinger T, Monaco AP 1994 Mutations in the DAX-1 gene give rise to both X-linked adrenal hypoplasia congenita and hypogonadotropic hypogonadism. Nature 372:672–676 - PubMed
-
- Tamai KT, Monaco L, Alastalo TP, Lalli E, Parvinen M, Sassone-Corsi P 1996 Hormonal and developmental regulation of DAX-1 expression in Sertoli cells. Mol Endocrinol 10:1561–1569 - PubMed
-
- Reutens AT, Achermann JC, Ito M, Gu WX, Habiby RL, Donohoue PA, Pang S, Hindmarsh PC, Jameson JL 1999 Clinical and functional effects of mutations in the DAX-1 gene in patients with adrenal hypoplasia congenita. J Clin Endocrinol Metab 84:504–511 - PubMed
-
- Meeks JJ, Weiss J, Jameson JL 2003 Dax1 is required for testis determination. Nat Genet 34:32–33 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

