Extracellular nucleotides stimulate Cl- currents in biliary epithelia through receptor-mediated IP3 and Ca2+ release
- PMID: 18787062
- PMCID: PMC2584822
- DOI: 10.1152/ajpgi.90382.2008
Extracellular nucleotides stimulate Cl- currents in biliary epithelia through receptor-mediated IP3 and Ca2+ release
Abstract
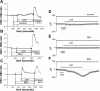
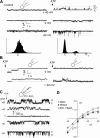
Extracellular ATP regulates bile formation by binding to P2 receptors on cholangiocytes and stimulating transepithelial Cl(-) secretion. However, the specific signaling pathways linking receptor binding to Cl(-) channel activation are not known. Consequently, the aim of these studies in human Mz-Cha-1 biliary cells and normal rat cholangiocyte monolayers was to assess the intracellular pathways responsible for ATP-stimulated increases in intracellular Ca(2+) concentration ([Ca(2+)](i)) and membrane Cl(-) permeability. Exposure of cells to ATP resulted in a rapid increase in [Ca(2+)](i) and activation of membrane Cl(-) currents; both responses were abolished by prior depletion of intracellular Ca(2+). ATP-stimulated Cl(-) currents demonstrated mild outward rectification, reversal at E(Cl(-)), and a single-channel conductance of approximately 17 pS, where E is the equilibrium potential. The conductance response to ATP was inhibited by the Cl(-) channel inhibitors NPPB and DIDS but not the CFTR inhibitor CFTR(inh)-172. Both ATP-stimulated increases in [Ca(2+)](i) and Cl(-) channel activity were inhibited by the P2Y receptor antagonist suramin. The PLC inhibitor U73122 and the inositol 1,4,5-triphosphate (IP3) receptor inhibitor 2-APB both blocked the ATP-stimulated increase in [Ca(2+)](i) and membrane Cl(-) currents. Intracellular dialysis with purified IP3 activated Cl(-) currents with identical properties to those activated by ATP. Exposure of normal rat cholangiocyte monolayers to ATP increased short-circuit currents (I(sc)), reflecting transepithelial secretion. The I(sc) was unaffected by CFTR(inh)-172 but was significantly inhibited by U73122 or 2-APB. In summary, these findings indicate that the apical P2Y-IP3 receptor signaling complex is a dominant pathway mediating biliary epithelial Cl(-) transport and, therefore, may represent a potential target for increasing secretion in the treatment of cholestatic liver disease.
Figures







Similar articles
-
Bile acids stimulate cholangiocyte fluid secretion by activation of transmembrane member 16A Cl- channels.Hepatology. 2018 Jul;68(1):187-199. doi: 10.1002/hep.29804. Epub 2018 May 9. Hepatology. 2018. PMID: 29360145 Free PMC article.
-
Identification and functional characterization of the intermediate-conductance Ca(2+)-activated K(+) channel (IK-1) in biliary epithelium.Am J Physiol Gastrointest Liver Physiol. 2009 Nov;297(5):G1009-18. doi: 10.1152/ajpgi.00223.2009. Am J Physiol Gastrointest Liver Physiol. 2009. PMID: 20501432 Free PMC article.
-
Endogenous ATP release regulates Cl- secretion in cultured human and rat biliary epithelial cells.Am J Physiol. 1999 Jun;276(6):G1391-400. doi: 10.1152/ajpgi.1999.276.6.G1391. Am J Physiol. 1999. PMID: 10362642
-
Control of epithelial transport via luminal P2 receptors.Am J Physiol Renal Physiol. 2003 Mar;284(3):F419-32. doi: 10.1152/ajprenal.00075.2002. Am J Physiol Renal Physiol. 2003. PMID: 12556361 Review.
-
Transport systems in cholangiocytes: their role in bile formation and cholestasis.Yale J Biol Med. 1997 Jul-Aug;70(4):427-34. Yale J Biol Med. 1997. PMID: 9626763 Free PMC article. Review.
Cited by
-
Calcium signaling and the secretory activity of bile duct epithelia.Cell Calcium. 2014 Jun;55(6):317-24. doi: 10.1016/j.ceca.2014.02.003. Epub 2014 Feb 12. Cell Calcium. 2014. PMID: 24612866 Free PMC article. Review.
-
Bile acids stimulate cholangiocyte fluid secretion by activation of transmembrane member 16A Cl- channels.Hepatology. 2018 Jul;68(1):187-199. doi: 10.1002/hep.29804. Epub 2018 May 9. Hepatology. 2018. PMID: 29360145 Free PMC article.
-
Pathobiology of biliary epithelia.Biochim Biophys Acta Mol Basis Dis. 2018 Apr;1864(4 Pt B):1220-1231. doi: 10.1016/j.bbadis.2017.06.024. Epub 2017 Jul 15. Biochim Biophys Acta Mol Basis Dis. 2018. PMID: 28716705 Free PMC article. Review.
-
Conjunctival Fluid Secretion Impairment via CaCC-CFTR Dysfunction Is the Key Mechanism in Environmental Dry Eye.Int J Mol Sci. 2022 Nov 19;23(22):14399. doi: 10.3390/ijms232214399. Int J Mol Sci. 2022. PMID: 36430877 Free PMC article.
-
P2Y2 purinergic receptor gene deletion protects mice from bacterial endotoxin and sepsis-associated liver injury and mortality.Am J Physiol Gastrointest Liver Physiol. 2023 Nov 1;325(5):G471-G491. doi: 10.1152/ajpgi.00090.2023. Epub 2023 Sep 12. Am J Physiol Gastrointest Liver Physiol. 2023. PMID: 37697947 Free PMC article.
References
-
- Abbracchio MP, Burnstock G, Boeynaems JM, Barnard EA, Boyer JL, Kennedy C, Knight GE, Fumagalli M, Gachet C, Jacobson KA, Weisman GA. International Union of Pharmacology LVIII: update on the P2Y G protein-coupled nucleotide receptors: from molecular mechanisms and pathophysiology to therapy. Pharmacol Rev 58: 281–341, 2006. - PMC - PubMed
-
- Alpini G, Roberts S, Kuntz SM, Ueno Y, Gubba S, Podila PV, Lesage G, LaRusso NF. Morphological, molecular, and functional heterogeneity of cholangiocytes from normal rat liver. Gastroenterology 110: 1636–1643, 1996. - PubMed
-
- Bai D, del Corsso C, Srinivas M, Spray DC. Block of specific gap junction channel subtypes by 2-aminoethoxydiphenyl borate (2-APB). J Pharmacol Exp Ther 319: 1452–1458, 2006. - PubMed
-
- Basavappa S, Middleton JP, Mangel A, McGill J, Cohn JA, Fitz JG. Cl− and K+ transport in human biliary cell lines. Gastroenterology 104: 1796–1805, 1993. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

