Oxidant-induced inhibition of the plasma membrane Ca2+-ATPase in pancreatic acinar cells: role of the mitochondria
- PMID: 18787078
- PMCID: PMC2584981
- DOI: 10.1152/ajpcell.00083.2008
Oxidant-induced inhibition of the plasma membrane Ca2+-ATPase in pancreatic acinar cells: role of the mitochondria
Abstract
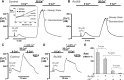
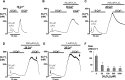
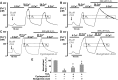
Impairment of the normal spatiotemporal pattern of intracellular Ca(2+) ([Ca(2+)](i)) signaling, and in particular, the transition to an irreversible "Ca(2+) overload" response, has been implicated in various pathophysiological states. In some diseases, including pancreatitis, oxidative stress has been suggested to mediate this Ca(2+) overload and the associated cell injury. We have previously demonstrated that oxidative stress with hydrogen peroxide (H(2)O(2)) evokes a Ca(2+) overload response and inhibition of plasma membrane Ca(2+)-ATPase (PMCA) in rat pancreatic acinar cells (Bruce JI and Elliott AC. Am J Physiol Cell Physiol 293: C938-C950, 2007). The aim of the present study was to further examine this oxidant-impaired inhibition of the PMCA, focusing on the role of the mitochondria. Using a [Ca(2+)](i) clearance assay in which mitochondrial Ca(2+) uptake was blocked with Ru-360, H(2)O(2) (50 microM-1 mM) markedly inhibited the PMCA activity. This H(2)O(2)-induced inhibition of the PMCA correlated with mitochondrial depolarization (assessed using tetramethylrhodamine methylester fluorescence) but could occur without significant ATP depletion (assessed using Magnesium Green fluorescence). The H(2)O(2)-induced PMCA inhibition was sensitive to the mitochondrial permeability transition pore (mPTP) inhibitors, cyclosporin-A and bongkrekic acid. These data suggest that oxidant-induced opening of the mPTP and mitochondrial depolarization may lead to an inhibition of the PMCA that is independent of mitochondrial Ca(2+) handling and ATP depletion, and we speculate that this may involve the release of a mitochondrial factor. Such a phenomenon may be responsible for the Ca(2+) overload response, and for the transition between apoptotic and necrotic cell death thought to be important in many disease states.
Figures



References
-
- Baggaley E, McLarnon S, Demeter I, Varga G, Bruce JI. Differential regulation of the apical plasma membrane Ca2+-ATPase by protein kinase A in parotid acinar cells. J Biol Chem 282: 37678–37693, 2007. - PubMed
-
- Baines CP, Kaiser RA, Purcell NH, Blair NS, Osinska H, Hambleton MA, Brunskill EW, Sayen MR, Gottlieb RA, Dorn GW, Robbins J, Molkentin JD. Loss of cyclophilin D reveals a critical role for mitochondrial permeability transition in cell death. Nature 434: 658–662, 2005. - PubMed
-
- Barros LF, Kanaseki T, Sabirov R, Morishima S, Castro J, Bittner CX, Maeno E, Ando-Akatsuka Y, Okada Y. Apoptotic and necrotic blebs in epithelial cells display similar neck diameters but different kinase dependency. Cell Death Differ 10: 687–697, 2003. - PubMed
-
- Barrow SL, Voronina SG, da Silva Xavier G, Chvanov MA, Longbottom RE, Gerasimenko OV, Petersen OH, Rutter GA, Tepikin AV. ATP depletion inhibits Ca2+ release, influx and extrusion in pancreatic acinar cells but not pathological Ca2+ responses induced by bile. Pflügers Arch 455: 1025–1039, 2008. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

