Protein trans-splicing as a means for viral vector-mediated in vivo gene therapy
- PMID: 18788906
- PMCID: PMC2940629
- DOI: 10.1089/hum.2008.009
Protein trans-splicing as a means for viral vector-mediated in vivo gene therapy
Abstract
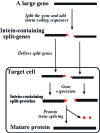
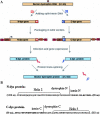
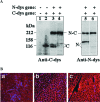
Inteins catalyze protein splicing in a fashion similar to how self-splicing introns catalyze RNA splicing. Splitinteins catalyze precise ligation of two separate polypeptides through trans-splicing in a highly specific manner. Here we report a method of using protein trans-splicing to circumvent the packaging size limit of gene therapy vectors. To demonstrate this method, we chose a large dystrophin gene and an adeno-associated viral (AAV) vector, which has a small packaging size. A highly functional 6.3-kb Becker-form dystrophin cDNA was broken into two pieces and modified by adding appropriate split-intein coding sequences, resulting in splitgenes sufficiently small for packaging in AAV vectors. The two split-genes, after codelivery into target cells, produced two polypeptides that spontaneously trans-spliced to form the expected Becker-form dystrophin protein in cell culture in vitro. Delivering the split-genes by AAV1 vectors into the muscle of a mouse model of Duchenne muscular dystrophy rendered therapeutic gene expression and benefits.
Figures
Similar articles
-
Expression of full-length dystrophin reverses muscular dystrophy defects in young and old mdx4cv mice.J Clin Invest. 2025 Jun 10;135(15):e189075. doi: 10.1172/JCI189075. eCollection 2025 Aug 1. J Clin Invest. 2025. PMID: 40493400 Free PMC article.
-
Triple trans-splicing adeno-associated virus vectors capable of transferring the coding sequence for full-length dystrophin protein into dystrophic mice.Hum Gene Ther. 2014 Feb;25(2):98-108. doi: 10.1089/hum.2013.164. Epub 2013 Dec 19. Hum Gene Ther. 2014. PMID: 24191945
-
Split intein-mediated protein trans-splicing to express large dystrophins.Nature. 2024 Aug;632(8023):192-200. doi: 10.1038/s41586-024-07710-8. Epub 2024 Jul 17. Nature. 2024. PMID: 39020181 Free PMC article.
-
[Gene therapy for muscular dystrophy].No To Hattatsu. 2004 Mar;36(2):117-23. No To Hattatsu. 2004. PMID: 15031985 Review. Japanese.
-
Perspective on Adeno-Associated Virus Capsid Modification for Duchenne Muscular Dystrophy Gene Therapy.Hum Gene Ther. 2015 Dec;26(12):786-800. doi: 10.1089/hum.2015.107. Epub 2015 Oct 15. Hum Gene Ther. 2015. PMID: 26414293 Free PMC article. Review.
Cited by
-
Therapeutic in vivo delivery of gene editing agents.Cell. 2022 Jul 21;185(15):2806-2827. doi: 10.1016/j.cell.2022.03.045. Epub 2022 Jul 6. Cell. 2022. PMID: 35798006 Free PMC article. Review.
-
Adeno-associated virus vector as a platform for gene therapy delivery.Nat Rev Drug Discov. 2019 May;18(5):358-378. doi: 10.1038/s41573-019-0012-9. Nat Rev Drug Discov. 2019. PMID: 30710128 Free PMC article. Review.
-
Broadening the reach and investigating the potential of prime editors through fully viral gene-deleted adenoviral vector delivery.Nucleic Acids Res. 2021 Nov 18;49(20):11986-12001. doi: 10.1093/nar/gkab938. Nucleic Acids Res. 2021. PMID: 34669958 Free PMC article.
-
Systemic delivery of full-length dystrophin in Duchenne muscular dystrophy mice.Nat Commun. 2024 Jul 21;15(1):6141. doi: 10.1038/s41467-024-50569-6. Nat Commun. 2024. PMID: 39034316 Free PMC article.
-
CRISPR-Based Tools for Fighting Rare Diseases.Life (Basel). 2022 Nov 24;12(12):1968. doi: 10.3390/life12121968. Life (Basel). 2022. PMID: 36556333 Free PMC article. Review.
References
-
- Chao H. Sun L. Bruce A. Xiao X. Walsh C.E. Expression of human factor VIII by splicing between dimerized AAV vectors. Mol. Ther. 2002;5:716–722. - PubMed
-
- Chen L. Zhu F. Li J. Lu H. Jiang H. Sarkar R. Arruda V.R. Wang J. Zhao J. Pierce G.F. Ding Q. Wang X. Wang H. Pipe S.W. Liu X.Q. Xiao X. Camire R.M. Xiao W. The enhancing effects of the light chain on heavy chain secretion in split delivery of factor VIII gene. Mol. Ther. 2007;15:1856–1862. - PMC - PubMed
-
- Duan D. Yue Y. Yan Z. Engelhardt J.F. A new dual-vector approach to enhance recombinant adeno-associated virus-mediated gene expression through intermolecular cis activation. Nat. Med. 2000;6:595–598. - PubMed
-
- Duan D. Yue Y. Engelhardt J.F. Expanding AAV packaging capacity with trans-splicing or overlapping vectors: A quantitative comparison. Mol. Ther. 2001;4:383–391. - PubMed
-
- England S.B. Nicholson L.V. Johnson M.A. Forrest S.M. Love D.R. Zubrzycka-Gaarn E.E. Bulman D.E. Harris J.B. Davies K.E. Very mild muscular dystrophy associated with the deletion of 46% of dystrophin. Nature. 1990;343:180–182. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

