Hydromorphone efficacy and treatment protocol impact on tolerance and mu-opioid receptor regulation
- PMID: 18789923
- PMCID: PMC2586831
- DOI: 10.1016/j.ejphar.2008.08.025
Hydromorphone efficacy and treatment protocol impact on tolerance and mu-opioid receptor regulation
Abstract
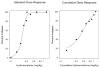
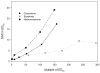
This study examined the antinociceptive (analgesic) efficacy of hydromorphone and hydromorphone-induced tolerance and regulation of mu-opioid receptor density. Initially s.c. hydromorphone's time of peak analgesic (tail-flick) effect (45 min) and ED50 using standard and cumulative dosing protocols (0.22 mg/kg, 0.37 mg/kg, respectively) were determined. The apparent analgesic efficacy (tau) of hydromorphone was then estimated using the operational model of agonism and the irreversible mu-opioid receptor antagonist clocinnamox. Mice were injected with clocinnamox (0.32-25.6 mg/kg, i.p.) and 24 h later, the analgesic potency of hydromorphone was determined. The tau value for hydromorphone was 35, which suggested that hydromorphone is a lower analgesic efficacy opioid agonist. To examine hydromorphone-induced tolerance, mice were continuously infused s.c. with hydromorphone (2.1-31.5 mg/kg/day) for 7 days and then morphine cumulative dose response studies were performed. Other groups of mice were injected with hydromorphone (2.2-22 mg/kg/day) once, or intermittently every 24 h for 7 days. Twenty-four hours after the last injection, mice were tested using morphine cumulative dosing studies. There was more tolerance with infusion treatments compared to intermittent treatment. When compared to higher analgesic efficacy opioids, hydromorphone infusions induced substantially more tolerance. Finally, the effect of chronic infusion (31.5 mg/kg/day) and 7 day intermittent (22 mg/kg/day) hydromorphone treatment on spinal cord mu-opioid receptor density was determined. Hydromorphone did not produce any change in mu-opioid receptor density following either treatment. These results support suggestions that analgesic efficacy is correlated with tolerance magnitude and regulation of mu-opioid receptors when opioid agonists are continuously administered. Taken together, these studies indicate that analgesic efficacy and treatment protocol are important in determining tolerance and regulation of mu-opioid receptors.
Figures




References
-
- Adams JU, Paronis CA, Holtzman SG. Assessment of relative intrinsic activity of mu–opioid analgesics in vivo by using beta-funaltrexamine. J Pharmacol Exp Ther. 1990;255:1027–1032. - PubMed
-
- Black JW, Leff P. Operational models of pharmacological agonism. Proc R Soc Lond B Biol Sci. 1983;220:141–162. - PubMed
-
- Bradford MM. A refined and sensitive method for the quantitation of microgram quantities of protein using the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. - PubMed
-
- Cicero TJ, Inciardi JA, Muñoz A. Trends in abuse of Oxycontin and other opioid analgesics in the United States: 2002–2004. J Pain. 2005;6:662–672. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

