Opposite effects of glucagon and insulin on compensation for spectacle lenses in chicks
- PMID: 18791176
- PMCID: PMC2755053
- DOI: 10.1167/iovs.08-1708
Opposite effects of glucagon and insulin on compensation for spectacle lenses in chicks
Abstract
Purpose: Chick eyes compensate for the defocus imposed by positive or negative spectacle lenses. Glucagon may signal the sign of defocus. Do insulin (or IGF-1) and glucagon act oppositely in controlling eye growth, as they do in metabolic pathways and in control of retinal neurogenesis?
Methods: Chicks, wearing lenses or diffusers or neither over both eyes, were injected with glucagon, a glucagon antagonist, insulin, or IGF-1 in one eye (saline in the other eye). Alternatively, chicks without lenses received insulin plus glucagon in one eye, and either glucagon or insulin in the fellow eye. Ocular dimensions, refractive errors, and glycosaminoglycan synthesis were measured over 2 to 4 days.
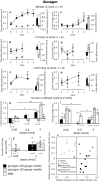
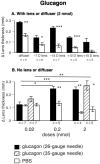
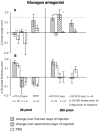
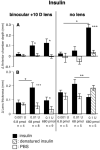
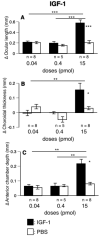
Results: Glucagon attenuated the myopic response to negative lenses or diffusers by slowing ocular elongation and thickening the choroid; in contrast, with positive lenses, it increased ocular elongation to normal levels and reduced choroidal thickening, as did a glucagon antagonist. Insulin prevented the hyperopic response to positive lenses by speeding ocular elongation and thinning the choroid. In eyes without lenses, both insulin and IGF-1 speeded, and glucagon slowed, ocular elongation, but glucagon and insulin each increased the rate of thickening of the crystalline lens. When injected together, insulin blocked choroidal thickening by glucagon, at a dose that did not, by itself, thin the choroid.
Conclusions: Glucagon and insulin (or IGF-1) cause generally opposite modulations of eye growth, with glucagon mostly increasing choroidal thickness and insulin mostly increasing ocular elongation. These effects are mutually inhibitory and depend on the visual input.
Figures


Similar articles
-
Choroidal and scleral mechanisms of compensation for spectacle lenses in chicks.Vision Res. 1995 May;35(9):1175-94. doi: 10.1016/0042-6989(94)00233-c. Vision Res. 1995. PMID: 7610579
-
Insulin acts as a powerful stimulator of axial myopia in chicks.Invest Ophthalmol Vis Sci. 2009 Jan;50(1):13-23. doi: 10.1167/iovs.08-1702. Epub 2008 Jul 3. Invest Ophthalmol Vis Sci. 2009. PMID: 18599564
-
Evidence for a potential role of glucagon during eye growth regulation in chicks.Vis Neurosci. 2002 Nov-Dec;19(6):755-66. doi: 10.1017/s0952523802196064. Vis Neurosci. 2002. PMID: 12688670
-
Temporal integration of visual signals in lens compensation (a review).Exp Eye Res. 2013 Sep;114:69-76. doi: 10.1016/j.exer.2013.02.014. Epub 2013 Mar 5. Exp Eye Res. 2013. PMID: 23470505 Free PMC article. Review.
-
The multifunctional choroid.Prog Retin Eye Res. 2010 Mar;29(2):144-68. doi: 10.1016/j.preteyeres.2009.12.002. Epub 2009 Dec 29. Prog Retin Eye Res. 2010. PMID: 20044062 Free PMC article. Review.
Cited by
-
The dynamic sclera: extracellular matrix remodeling in normal ocular growth and myopia development.Exp Eye Res. 2015 Apr;133:100-11. doi: 10.1016/j.exer.2014.07.015. Exp Eye Res. 2015. PMID: 25819458 Free PMC article. Review.
-
Correlation analysis of myopia and dietary factors among primary and secondary school students in Shenyang, China.Sci Rep. 2024 Sep 4;14(1):20619. doi: 10.1038/s41598-024-71254-0. Sci Rep. 2024. PMID: 39232029 Free PMC article.
-
Effects of intravitreal insulin and insulin signaling cascade inhibitors on emmetropization in the chick.Mol Vis. 2012;18:2608-22. Epub 2012 Oct 20. Mol Vis. 2012. PMID: 23112573 Free PMC article.
-
The choroid-sclera interface: An ultrastructural study.Heliyon. 2022 May 10;8(5):e09408. doi: 10.1016/j.heliyon.2022.e09408. eCollection 2022 May. Heliyon. 2022. PMID: 35586330 Free PMC article.
-
IGF-1 gene polymorphisms in Polish families with high-grade myopia.Mol Vis. 2011;17:2428-39. Epub 2011 Sep 21. Mol Vis. 2011. PMID: 21976954 Free PMC article.
References
-
- Wildsoet CF. Active emmetropization--evidence for its existence and ramifications for clinical practice. Ophthalmic Physiol Opt. 1997;17:279–90. - PubMed
-
- Wallman J, Winawer J. Homeostasis of eye growth and the question of myopia. Neuron. 2004;43:447–68. - PubMed
-
- Fischer AJ, McGuire JJ, Schaeffel F, Stell WK. Light- and focus-dependent expression of the transcription factor ZENK in the chick retina. Nat Neurosci. 1999;2:706–12. - PubMed
-
- Bitzer M, Schaeffel F. Defocus-induced changes in ZENK expression in the chicken retina. Invest Ophthal Vis Sci. 2002;43:246–52. - PubMed
-
- Feldkaemper MP, Wang HY, Schaeffel F. Changes in retinal and choroidal gene expression during development of refractive errors in chicks. Invest Ophthal Vis Sci. 2000;41:1623–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

