Control of cellular GADD34 levels by the 26S proteasome
- PMID: 18794359
- PMCID: PMC2593369
- DOI: 10.1128/MCB.00724-08
Control of cellular GADD34 levels by the 26S proteasome
Abstract
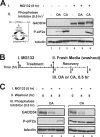
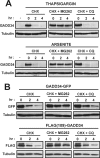
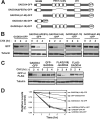
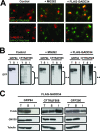
GADD34, the product of a growth arrest and DNA damage-inducible gene, is expressed at low levels in unstressed cells. In response to stress, the cellular content of GADD34 protein increases and, on termination of stress, rapidly declines. We investigated the mechanisms that control GADD34 levels in human cells. GADD34 proteins containing either an internal FLAG or a C-terminal green fluorescent protein epitope were degraded at rates similar to endogenous GADD34. However, the addition of epitopes at the N terminus or deletion of N-terminal sequences stabilized GADD34. N-terminal peptides of GADD34, either alone or fused to heterologous proteins, exhibited rapid degradation similar to wild-type GADD34, thereby identifying an N-terminal degron. Deletion of internal PEST repeats had no impact on GADD34 stability but modulated the binding and activity of protein phosphatase 1. Proteasomal but not lysosomal inhibitors enhanced GADD34 stability and eukaryotic initiation factor 2alpha (eIF-2alpha) dephosphorylation, a finding consistent with GADD34's role in assembling an eIF-2alpha phosphatase. GADD34 was polyubiquitinated, and this modification enhanced its turnover in cells. A stabilized form of GADD34 promoted the accumulation and aggregation of the mutant cystic fibrosis transmembrane conductance regulator (CFTRDeltaF508), highlighting the physiological importance of GADD34 turnover in protein processing in the endoplasmic reticulum and the potential impact of prolonged GADD34 expression in human disease.
Figures




References
-
- Akabas, M. H. 2000. Cystic fibrosis transmembrane conductance regulator: structure and function of an epithelial chloride channel. J. Biol. Chem. 2753729-3732. - PubMed
-
- Bence, N. F., R. M. Sampat, and R. R. Kopito. 2001. Impairment of the ubiquitin-proteasome system by protein aggregation. Science 2921552-1555. - PubMed
-
- Brush, M. H., D. C. Weiser, and S. Shenolikar. 2003. Growth arrest and DNA damage-inducible protein, GADD34, targets protein phosphatase 1 to the endoplasmic reticulum and promotes dephosphorylation of α-subunit of eukaryotic translation initiation factor 2. Mol. Cell. Biol. 231292-1303. - PMC - PubMed
-
- Ciechanover, A., and R. Ben-Saadon. 2004. N-terminal ubiquitination: more protein substrates join in. Trends Cell Biol. 14103-106. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
