Identification of Tspan9 as a novel platelet tetraspanin and the collagen receptor GPVI as a component of tetraspanin microdomains
- PMID: 18795891
- PMCID: PMC2652832
- DOI: 10.1042/BJ20081126
Identification of Tspan9 as a novel platelet tetraspanin and the collagen receptor GPVI as a component of tetraspanin microdomains
Abstract
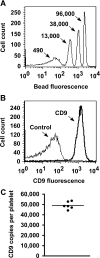
Platelets are essential for wound healing and inflammatory processes, but can also play a deleterious role by causing heart attack and stroke. Normal platelet activation is dependent on tetraspanins, a superfamily of glycoproteins that function as 'organisers' of cell membranes by recruiting other receptors and signalling proteins into tetraspanin-enriched microdomains. However, our understanding of how tetraspanin microdomains regulate platelets is hindered by the fact that only four of the 33 mammalian tetraspanins have been identified in platelets. This is because of a lack of antibodies to most tetraspanins and difficulties in measuring mRNA, due to low levels in this anucleate cell. To identify potentially platelet-expressed tetraspanins, mRNA was measured in their nucleated progenitor cell, the megakaryocyte, using serial analysis of gene expression and DNA microarrays. Amongst 19 tetraspanins identified in megakaryocytes, Tspan9, a previously uncharacterized tetraspanin, was relatively specific to these cells. Through generating the first Tspan9 antibodies, Tspan9 expression was found to be tightly regulated in platelets. The relative levels of CD9, CD151, Tspan9 and CD63 were 100, 14, 6 and 2 respectively. Since CD9 was expressed at 49000 cell surface copies per platelet, this suggested a copy number of 2800 Tspan9 molecules. Finally, Tspan9 was shown to be a component of tetraspanin microdomains that included the collagen receptor GPVI (glycoprotein VI) and integrin alpha6beta1, but not the von Willebrand receptor GPIbalpha or the integrins alphaIIbbeta3 or alpha2beta1. These findings suggest a role for Tspan9 in regulating platelet function in concert with other platelet tetraspanins and their associated proteins.
Figures

Similar articles
-
Tetraspanin Tspan9 regulates platelet collagen receptor GPVI lateral diffusion and activation.Platelets. 2017 Nov;28(7):629-642. doi: 10.1080/09537104.2016.1254175. Epub 2016 Dec 29. Platelets. 2017. PMID: 28032533 Free PMC article.
-
Palmitoylation supports the association of tetraspanin CD63 with CD9 and integrin alphaIIbbeta3 in activated platelets.Thromb Res. 2010 Feb;125(2):152-8. doi: 10.1016/j.thromres.2009.07.005. Epub 2009 Jul 29. Thromb Res. 2010. PMID: 19640571
-
Platelet tetraspanin complexes and their association with lipid rafts.Thromb Haemost. 2007 Nov;98(5):1081-7. Thromb Haemost. 2007. PMID: 18000614
-
The emerging role of tetraspanin microdomains on endothelial cells.Biochem Soc Trans. 2011 Dec;39(6):1667-73. doi: 10.1042/BST20110745. Biochem Soc Trans. 2011. PMID: 22103505 Review.
-
Tetraspanin microdomains: fine-tuning platelet function.Biochem Soc Trans. 2011 Apr;39(2):518-23. doi: 10.1042/BST0390518. Biochem Soc Trans. 2011. PMID: 21428931 Review.
Cited by
-
Identification of the tetraspanin CD82 as a new barrier to xenotransplantation.J Immunol. 2013 Sep 1;191(5):2796-805. doi: 10.4049/jimmunol.1300601. Epub 2013 Jul 19. J Immunol. 2013. PMID: 23872050 Free PMC article.
-
Transcriptome Profiling Reveals PHLDA1 as a Novel Molecular Marker for Ischemic Cardiomyopathy.J Mol Neurosci. 2018 May;65(1):102-109. doi: 10.1007/s12031-018-1066-6. Epub 2018 May 8. J Mol Neurosci. 2018. PMID: 29736818 Free PMC article.
-
Intracoronary Imaging, Cholesterol Efflux, and Transcriptomics after Intensive Statin Treatment in Diabetes.Sci Rep. 2017 Aug 1;7(1):7001. doi: 10.1038/s41598-017-07029-7. Sci Rep. 2017. PMID: 28765529 Free PMC article.
-
TspanC8 Tetraspanins and A Disintegrin and Metalloprotease 10 (ADAM10) Interact via Their Extracellular Regions: EVIDENCE FOR DISTINCT BINDING MECHANISMS FOR DIFFERENT TspanC8 PROTEINS.J Biol Chem. 2016 Feb 12;291(7):3145-57. doi: 10.1074/jbc.M115.703058. Epub 2015 Dec 14. J Biol Chem. 2016. PMID: 26668317 Free PMC article.
-
Tetraspanin Tspan9 regulates platelet collagen receptor GPVI lateral diffusion and activation.Platelets. 2017 Nov;28(7):629-642. doi: 10.1080/09537104.2016.1254175. Epub 2016 Dec 29. Platelets. 2017. PMID: 28032533 Free PMC article.
References
-
- Langer H. F., Gawaz M. Platelet-vessel wall interactions in atherosclerotic disease. Thromb. Haemost. 2008;99:480–486. - PubMed
-
- Varga-Szabo D., Pleines I., Nieswandt B. Cell adhesion mechanisms in platelets. Arterioscler. Thromb. Vasc. Biol. 2008;28:403–412. - PubMed
-
- Jacobson K., Mouritsen O. G., Anderson R. G. Lipid rafts: at a crossroad between cell biology and physics. Nat. Cell Biol. 2007;9:7–14. - PubMed
-
- Hemler M. E. Tetraspanin functions and associated microdomains. Nat. Rev. Mol. Cell Biol. 2005;6:801–811. - PubMed
-
- Le Naour F., Andre M., Boucheix C., Rubinstein E. Membrane microdomains and proteomics: lessons from tetraspanin microdomains and comparison with lipid rafts. Proteomics. 2006;6:6447–6454. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

