Constraint-induced movement therapy in the adult rat after unilateral corticospinal tract injury
- PMID: 18799672
- PMCID: PMC6671131
- DOI: 10.1523/JNEUROSCI.1697-08.2008
Constraint-induced movement therapy in the adult rat after unilateral corticospinal tract injury
Abstract
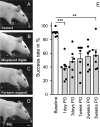
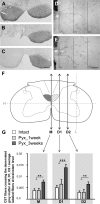
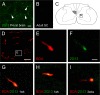
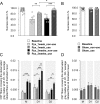
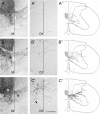
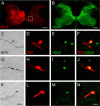
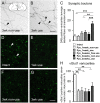
Smaller spinal cord injuries often allow some degree of spontaneous behavioral improvements because of structural rearrangements within different descending fiber tracts or intraspinal circuits. In this study, we investigate whether rehabilitative training of the forelimb (forced limb use) influences behavioral recovery and plastic events after injury to a defined spinal tract, the corticospinal tract (CST). Female adult Lewis rats received a unilateral CST injury at the brainstem level. Use of the contralateral impaired forelimb was either restricted, by a cast, or forced, by casting the unimpaired forelimb immediately after injury for either 1 or 3 weeks. Forced use of the impaired forelimb was followed by full behavioral recovery on the irregular horizontal ladder, whereas animals that could not use their affected side remained impaired. BDA (biotinylated dextran amine) labeling of the intact CST showed lesion-induced growth across the midline where CST collaterals increased their innervation density and extended fibers toward the ventral and the dorsal horn in response to forced limb use. Gene chip analysis of the denervated ventral horn revealed changes in particular for growth factors, adhesion and guidance molecules, as well as components of synapse formation suggesting an important role for these factors in activity-dependent intraspinal reorganization after unilateral CST injury.
Figures


Similar articles
-
Bilateral cervical contusion spinal cord injury in rats.Exp Neurol. 2009 Nov;220(1):9-22. doi: 10.1016/j.expneurol.2009.06.012. Epub 2009 Jun 25. Exp Neurol. 2009. PMID: 19559699 Free PMC article.
-
Regenerative growth of corticospinal tract axons via the ventral column after spinal cord injury in mice.J Neurosci. 2008 Jul 2;28(27):6836-47. doi: 10.1523/JNEUROSCI.5372-07.2008. J Neurosci. 2008. PMID: 18596159 Free PMC article.
-
Reticulospinal plasticity after cervical spinal cord injury in the rat involves withdrawal of projections below the injury.Exp Neurol. 2013 Sep;247:241-9. doi: 10.1016/j.expneurol.2013.05.003. Epub 2013 May 17. Exp Neurol. 2013. PMID: 23684634
-
Corticospinal reorganization after spinal cord injury.J Physiol. 2012 Aug 15;590(16):3647-63. doi: 10.1113/jphysiol.2012.233189. Epub 2012 May 14. J Physiol. 2012. PMID: 22586214 Free PMC article. Review.
-
Alternative routes for recovery of hand functions after corticospinal tract injury in primates and rodents.Curr Opin Neurol. 2019 Dec;32(6):836-843. doi: 10.1097/WCO.0000000000000749. Curr Opin Neurol. 2019. PMID: 31688166 Review.
Cited by
-
Dipolar cortico-muscular electrical stimulation: a novel method that enhances motor function in both - normal and spinal cord injured mice.J Neuroeng Rehabil. 2010 Sep 17;7:46. doi: 10.1186/1743-0003-7-46. J Neuroeng Rehabil. 2010. PMID: 20849604 Free PMC article.
-
Oligodendrocytic but not neuronal Nogo restricts corticospinal axon sprouting after CNS injury.Exp Neurol. 2018 Nov;309:32-43. doi: 10.1016/j.expneurol.2018.07.013. Epub 2018 Jul 25. Exp Neurol. 2018. PMID: 30055160 Free PMC article.
-
Extensive spontaneous plasticity of corticospinal projections after primate spinal cord injury.Nat Neurosci. 2010 Dec;13(12):1505-10. doi: 10.1038/nn.2691. Epub 2010 Nov 14. Nat Neurosci. 2010. PMID: 21076427 Free PMC article.
-
PTEN deletion enhances the regenerative ability of adult corticospinal neurons.Nat Neurosci. 2010 Sep;13(9):1075-81. doi: 10.1038/nn.2603. Epub 2010 Aug 8. Nat Neurosci. 2010. PMID: 20694004 Free PMC article.
-
Cortical overexpression of neuronal calcium sensor-1 induces functional plasticity in spinal cord following unilateral pyramidal tract injury in rat.PLoS Biol. 2010 Jun 22;8(6):e1000399. doi: 10.1371/journal.pbio.1000399. PLoS Biol. 2010. PMID: 20585375 Free PMC article.
References
-
- Adams JC. Biotin amplification of biotin and horseradish peroxidase signals in histochemical stains. J Histochem Cytochem. 1992;40:1457–1463. - PubMed
-
- Barbeau H, Rossignol S. Enhancement of locomotor recovery following spinal cord injury. Curr Opin Neurol. 1994;7:517–524. - PubMed
-
- Bareyre FM, Schwab ME. Inflammation, degeneration and regeneration in the injured spinal cord: insights from DNA microarrays. Trends Neurosci. 2003;26:555–563. - PubMed
-
- Bareyre FM, Kerschensteiner M, Raineteau O, Mettenleiter TC, Weinmann O, Schwab ME. The injured spinal cord spontaneously forms a new intraspinal circuit in adult rats. Nat Neurosci. 2004;7:269–277. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
