Sorbitol can fuel mouse sperm motility and protein tyrosine phosphorylation via sorbitol dehydrogenase
- PMID: 18799757
- PMCID: PMC2804811
- DOI: 10.1095/biolreprod.108.068882
Sorbitol can fuel mouse sperm motility and protein tyrosine phosphorylation via sorbitol dehydrogenase
Abstract
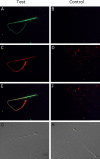
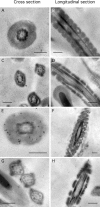
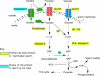
Energy sources that can be metabolized to yield ATP are essential for normal sperm functions such as motility. Two major monosaccharides, sorbitol and fructose, are present in semen. Furthermore, sorbitol dehydrogenase (SORD) can convert sorbitol to fructose, which can then be metabolized via the glycolytic pathway in sperm to make ATP. Here we characterize Sord mRNA and SORD expression during mouse spermatogenesis and examine the ability of sorbitol to support epididymal sperm motility and tyrosine phosphorylation. Sord mRNA levels increased during the course of spermatogenic differentiation. SORD protein, however, was first detected at the condensing spermatid stage. By indirect immunofluorescence, SORD was present along the length of the flagella of caudal epididymal sperm. Furthermore, immunoelectron microscopy showed that SORD was associated with mitochondria and the plasma membranes of sperm. Sperm incubated with sorbitol maintained motility, indicating that sorbitol was utilized as an energy source. Sorbitol, as well as glucose and fructose, were not essential to induce hyperactive motility. Protein tyrosine phosphorylation increased in a similar manner when sorbitol was substituted for glucose in the incubation medium used for sperm capacitation. These results indicate that sorbitol can serve as an alternative energy source for sperm motility and protein tyrosine phosphorylation.
Figures





Similar articles
-
Protein profile screening: reduced expression of Sord in the mouse epididymis induced by nicotine inhibits tyrosine phosphorylation level in capacitated spermatozoa.Reproduction. 2016 Mar;151(3):227-37. doi: 10.1530/REP-15-0370. Epub 2015 Dec 8. Reproduction. 2016. PMID: 26647419
-
Polyol pathway along the bovine epididymis.Mol Reprod Dev. 2004 Dec;69(4):448-56. doi: 10.1002/mrd.20170. Mol Reprod Dev. 2004. PMID: 15457514
-
Electrophoretic pattern of sorbitol dehydrogenase (E.C.1.1.1.14) in human seminal plasma and spermatozoa.Ann Genet. 1989;32(1):33-5. Ann Genet. 1989. PMID: 2751245
-
Protein phosphorylation in mammalian spermatozoa.Reproduction. 2003 Jan;125(1):17-26. doi: 10.1530/rep.0.1250017. Reproduction. 2003. PMID: 12622692 Review.
-
Signaling mechanisms in mammalian sperm motility.Biol Reprod. 2017 Jan 1;96(1):2-12. doi: 10.1095/biolreprod.116.144337. Biol Reprod. 2017. PMID: 28395326 Review.
Cited by
-
Protamine-2 Deficiency Initiates a Reactive Oxygen Species (ROS)-Mediated Destruction Cascade during Epididymal Sperm Maturation in Mice.Cells. 2020 Jul 27;9(8):1789. doi: 10.3390/cells9081789. Cells. 2020. PMID: 32727081 Free PMC article.
-
Efficacy and mechanism of lipoic acid in the treatment of reproductive injury caused by perfluorooctanoic acid.Exp Ther Med. 2023 Jan 30;25(3):116. doi: 10.3892/etm.2023.11815. eCollection 2023 Mar. Exp Ther Med. 2023. PMID: 36815965 Free PMC article.
-
Mitochondrial and cytoskeletal alterations are involved in the pathogenesis of hydronephrosis in ICR/Mlac-hydro mice.Int J Clin Exp Med. 2015 Jun 15;8(6):9192-204. eCollection 2015. Int J Clin Exp Med. 2015. PMID: 26309577 Free PMC article.
-
Nutritional aphrodisiacs: Biochemistry and Pharmacology.Curr Res Food Sci. 2024 Jun 11;9:100783. doi: 10.1016/j.crfs.2024.100783. eCollection 2024. Curr Res Food Sci. 2024. PMID: 38974844 Free PMC article. Review.
-
A systematic analysis of a deep mouse epididymal sperm proteome.Biol Reprod. 2012 Dec 21;87(6):141. doi: 10.1095/biolreprod.112.104208. Print 2012 Jun. Biol Reprod. 2012. PMID: 23115268 Free PMC article.
References
-
- Palomo MJ, FernAndez-Novell JM, Pena A, Guinovart JJ, Rigau T, Rodriguez-Gil JE.Glucose- and fructose-induced dog-sperm glycogen synthesis shows specific changes in the location of the sperm glycogen deposition. Mol Reprod Dev 2003; 64: 349–359. - PubMed
-
- O'Shea T, Wales RG.Metabolism of sorbitol and fructose by ram spermatozoa. J Reprod Fertil 1965; 10: 353–368. - PubMed
-
- Jones AR, Connor DE.Fructose metabolism by mature boar spermatozoa. Reprod Fertil Dev 2000; 12: 355–359. - PubMed
-
- Mann T, White IG.Metabolism of glycerol, sorbitol and related compounds by spermatozoa. Nature 1956; 178: 142–143. - PubMed
-
- Medrano A, Fernandez-Novell JM, Ramio L, Alvarez J, Goldberg E, Montserrat Rivera M, Guinovart JJ, Rigau T, Rodriguez-Gil JE.Utilization of citrate and lactate through a lactate dehydrogenase and ATP-regulated pathway in boar spermatozoa. Mol Reprod Dev 2006; 73: 369–378. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

