Conformational changes and flexibility in T-cell receptor recognition of peptide-MHC complexes
- PMID: 18800968
- PMCID: PMC2782316
- DOI: 10.1042/BJ20080850
Conformational changes and flexibility in T-cell receptor recognition of peptide-MHC complexes
Abstract
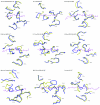
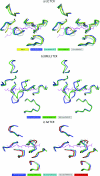
A necessary feature of the immune system, TCR (T-cell receptor) cross-reactivity has been implicated in numerous autoimmune pathologies and is an underlying cause of transplant rejection. Early studies of the interactions of alphabeta TCRs (T-cell receptors) with their peptide-MHC ligands suggested that conformational plasticity in the TCR CDR (complementarity determining region) loops is a dominant contributor to T-cell cross-reactivity. Since these initial studies, the database of TCRs whose structures have been solved both bound and free is now large enough to permit general conclusions to be drawn about the extent of TCR plasticity and the types and locations of motion that occur. In the present paper, we review the conformational differences between free and bound TCRs, quantifying the structural changes that occur and discussing their possible roles in specificity and cross-reactivity. We show that, rather than undergoing major structural alterations or 'folding' upon binding, the majority of TCR CDR loops shift by relatively small amounts. The structural changes that do occur are dominated by hinge-bending motions, with loop remodelling usually occurring near loop apexes. As predicted from previous studies, the largest changes are in the hypervariable CDR3alpha and CDR3beta loops, although in some cases the germline-encoded CDR1alpha and CDR2alpha loops shift in magnitudes that approximate those of the CDR3 loops. Intriguingly, the smallest shifts are in the germline-encoded loops of the beta-chain, consistent with recent suggestions that the TCR beta domain may drive ligand recognition.
Figures


References
-
- Mason D. A very high level of crossreactivity is an essential feature of the T-cell receptor. Immunol. Today. 1998;19:395–404. - PubMed
-
- Wilson D. B., Wilson D. H., Schroder K., Pinilla C., Blondelle S., Houghten R. A., Garcia K. C. Specificity and degeneracy of T cells. Mol. Immunol. 2004;40:1047–1055. - PubMed
-
- Maverakis E., van den Elzen P., Sercarz E. E. Self-reactive T cells and degeneracy of T cell recognition: evolving concepts–from sequence homology to shape mimicry and TCR flexibility. J. Autoimmun. 2001;16:201–209. - PubMed
-
- Garcia K. C., Adams E. J. How the T cell receptor sees antigen – a structural view. Cell. 2005;122:333–336. - PubMed
-
- Rudolph M. G., Stanfield R. L., Wilson I. A. How TCRs bind MHCs, peptides, and coreceptors. Annu. Rev. Immunol. 2006;24:419–466. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

