Acetogenesis and the Wood-Ljungdahl pathway of CO(2) fixation
- PMID: 18801467
- PMCID: PMC2646786
- DOI: 10.1016/j.bbapap.2008.08.012
Acetogenesis and the Wood-Ljungdahl pathway of CO(2) fixation
Abstract
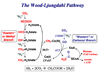
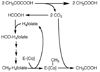
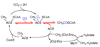
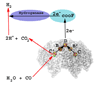
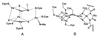
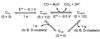
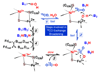
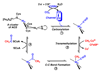
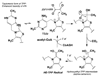
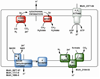
Conceptually, the simplest way to synthesize an organic molecule is to construct it one carbon at a time. The Wood-Ljungdahl pathway of CO(2) fixation involves this type of stepwise process. The biochemical events that underlie the condensation of two one-carbon units to form the two-carbon compound, acetate, have intrigued chemists, biochemists, and microbiologists for many decades. We begin this review with a description of the biology of acetogenesis. Then, we provide a short history of the important discoveries that have led to the identification of the key components and steps of this usual mechanism of CO and CO(2) fixation. In this historical perspective, we have included reflections that hopefully will sketch the landscape of the controversies, hypotheses, and opinions that led to the key experiments and discoveries. We then describe the properties of the genes and enzymes involved in the pathway and conclude with a section describing some major questions that remain unanswered.
Figures








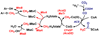


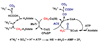

References
-
- Drake HL, Daniel SL, Matthies C, Küsel K. Acetogenesis, acetogenic bacteria, and the acetyl-CoA pathway: Past and current perspectives. In: Drake HL, editor. Acetogenesis. New York: Chapman and Hall; 1994. pp. 3–60.
-
- Müller V, Imkamp F, Rauwolf Andreas, Küsel K, Drake HL. Molecular and Cellular Biology of Acetogenic Bacteria. In: Nakano MM, Zuber P, editors. Strict and Facultative Anaerobes: Medical and Environmental Aspects. Wymondham, UK: Horizon Bioscience; 2004. pp. 251–281.
-
- Fischer F, Lieske R, Winzer K. Biologische gasreaktionen. II. Gber die bildung von essigs ure bei der biologischen umsetzung von kohlenoxyd und kohlens ure mit wasserstoff zu methan. Biochem. Z. 1932;245:2–12.
-
- Wieringa KT. Over het verdwinjhnen van waterstof en koolzuur onder anaerobe voorwaarden. Antonie van Leeuwenhoek. 1936;3:263–273.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

