Tissue-specific difference in the molecular mechanisms for the development of acute insulin resistance after injury
- PMID: 18801909
- PMCID: PMC2630903
- DOI: 10.1210/en.2008-0742
Tissue-specific difference in the molecular mechanisms for the development of acute insulin resistance after injury
Abstract
Acute insulin resistance occurs after injury, hemorrhage, infection, and critical illness. However, little is known about the development of this acute insulin-resistant state. In the current study, we found that insulin resistance develops rapidly in skeletal muscle, with the earliest insulin signaling defects at 60 min. However, defects in insulin signaling were measurable even earlier in liver, by as soon as 15 min after hemorrhage. To begin to understand the mechanisms for the development of acute insulin resistance, serine phosphorylation of insulin receptor substrate (IRS)-1 and c-Jun N-terminal kinase phosphorylation/activation was investigated. These markers (and possible contributors) of insulin resistance were increased in the liver after hemorrhage but not measurable in skeletal muscle. Because glucocorticoids are important counterregulatory hormones responsible for glucose homeostasis, a glucocorticoid synthesis inhibitor, metyrapone, and a glucocorticoid receptor antagonist, RU486, were administered to adult rats prior to hemorrhage. In the liver, the defects of insulin signaling after hemorrhage, including reduced tyrosine phosphorylation of the insulin receptor and IRS-1, association between IRS-1 and phosphatidylinositol 3-kinase and serine phosphorylation of Akt in response to insulin were not altered by pretreatment of rats with metyrapone or RU486. In contrast, hemorrhage-induced defects in insulin signaling were dramatically reversed in skeletal muscle, indicating a prevention of insulin resistance in muscle. These results suggest that distinct mechanisms for hemorrhage-induced acute insulin resistance are present in these two tissues and that glucocorticoids are involved in the rapid development of insulin resistance in skeletal muscle, but not in the liver, after hemorrhage.
Figures
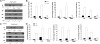






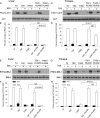
References
-
- Desouza C, Gilling L, Fonseca V 2001 Management of the insulin resistance syndrome. Curr Diab Rep 1:140–147 - PubMed
-
- Kendall DM, Harmel AP 2002 The metabolic syndrome, type 2 diabetes, and cardiovascular disease: understanding the role of insulin resistance. Am J Manag Care 8:S635–S653 - PubMed
-
- Ikezu T, Okamoto T, Yonezawa K, Tompkins RG, Martyn JA 1997 Analysis of thermal injury-induced insulin resistance in rodents. Implication of postreceptor mechanisms. J Biol Chem 272:25289–25295 - PubMed
-
- Carter EA 1998 Insulin resistance in burns and trauma. Nutr Rev 56:S170–S176 - PubMed
-
- Lange MP, Dahn MS, Jacobs LA 1985 The significance of hyperglycemia after injury. Heart Lung 14:470–472 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

