Mast cells promote airway smooth muscle cell differentiation via autocrine up-regulation of TGF-beta 1
- PMID: 18802103
- PMCID: PMC3992381
- DOI: 10.4049/jimmunol.181.7.5001
Mast cells promote airway smooth muscle cell differentiation via autocrine up-regulation of TGF-beta 1
Abstract
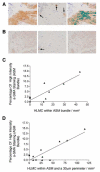
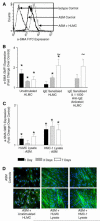
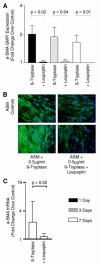
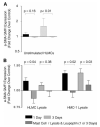
Asthma is a major cause of morbidity and mortality worldwide. It is characterized by airway dysfunction and inflammation. A key determinant of the asthma phenotype is infiltration of airway smooth muscle bundles by activated mast cells. We hypothesized that interactions between these cells promotes airway smooth muscle differentiation into a more contractile phenotype. In vitro coculture of human airway smooth muscle cells with beta-tryptase, or mast cells with or without IgE/anti-IgE activation, increased airway smooth muscle-derived TGF-beta1 secretion, alpha-smooth muscle actin expression and agonist-provoked contraction. This promotion to a more contractile phenotype was inhibited by both the serine protease inhibitor leupeptin and TGF-beta1 neutralization, suggesting that the observed airway smooth muscle differentiation was driven by the autocrine release of TGF-beta1 in response to activation by mast cell beta-tryptase. Importantly, in vivo we found that in bronchial mucosal biopsies from asthmatics the intensity of alpha-smooth muscle actin expression was strongly related to the number of mast cells within or adjacent to an airway smooth muscle bundle. These findings suggest that mast cell localization in the airway smooth muscle bundle promotes airway smooth muscle cell differentiation into a more contractile phenotype, thus contributing to the disordered airway physiology that characterizes asthma.
Figures



Similar articles
-
Caveolin-1 is required for contractile phenotype expression by airway smooth muscle cells.J Cell Mol Med. 2011 Nov;15(11):2430-42. doi: 10.1111/j.1582-4934.2010.01246.x. J Cell Mol Med. 2011. PMID: 21199324 Free PMC article.
-
Mast cell-airway smooth muscle crosstalk: the role of thymic stromal lymphopoietin.Chest. 2012 Jul;142(1):76-85. doi: 10.1378/chest.11-1782. Chest. 2012. PMID: 22052771 Free PMC article.
-
Tryptase-stimulated human airway smooth muscle cells induce cytokine synthesis and mast cell chemotaxis.FASEB J. 2003 Nov;17(14):2139-41. doi: 10.1096/fj.03-0041fje. Epub 2003 Sep 18. FASEB J. 2003. PMID: 14500550
-
The role of the mast cell in the pathophysiology of asthma.J Allergy Clin Immunol. 2006 Jun;117(6):1277-84. doi: 10.1016/j.jaci.2006.02.039. Epub 2006 Apr 27. J Allergy Clin Immunol. 2006. PMID: 16750987 Review.
-
The role of the mast cell in asthma: a reassessment.Curr Opin Allergy Clin Immunol. 2003 Feb;3(1):45-50. doi: 10.1097/00130832-200302000-00008. Curr Opin Allergy Clin Immunol. 2003. PMID: 12582314 Review.
Cited by
-
Mast cells in allergic asthma and beyond.Yonsei Med J. 2010 Nov;51(6):797-807. doi: 10.3349/ymj.2010.51.6.797. Yonsei Med J. 2010. PMID: 20879044 Free PMC article. Review.
-
Human bone marrow-mesenchymal stem cell-derived exosomal microRNA-188 reduces bronchial smooth muscle cell proliferation in asthma through suppressing the JARID2/Wnt/β-catenin axis.Cell Cycle. 2022 Feb;21(4):352-367. doi: 10.1080/15384101.2021.2020432. Epub 2022 Jan 2. Cell Cycle. 2022. PMID: 34974799 Free PMC article.
-
CADM1 controls actin cytoskeleton assembly and regulates extracellular matrix adhesion in human mast cells.PLoS One. 2014 Jan 22;9(1):e85980. doi: 10.1371/journal.pone.0085980. eCollection 2014. PLoS One. 2014. PMID: 24465823 Free PMC article.
-
IL-33 drives airway hyper-responsiveness through IL-13-mediated mast cell: airway smooth muscle crosstalk.Allergy. 2015 May;70(5):556-67. doi: 10.1111/all.12593. Epub 2015 Mar 16. Allergy. 2015. PMID: 25683166 Free PMC article.
-
Novel airway smooth muscle-mast cell interactions and a role for the TRPV4-ATP axis in non-atopic asthma.Eur Respir J. 2020 Jul 2;56(1):1901458. doi: 10.1183/13993003.01458-2019. Print 2020 Jul. Eur Respir J. 2020. PMID: 32299856 Free PMC article.
References
-
- Hartert TV, Peebles RS., Jr Epidemiology of asthma: the year in review. Curr. Opin. Pulm. Med. 2000;6:4–9. - PubMed
-
- Kay AB. Pathology of mild, severe, and fatal asthma. Am. J. Respir. Crit Care Med. 1996;154:S66–S69. - PubMed
-
- Wardlaw AJ, Brightling C, Green R, Woltmann G, Pavord I. Eosinophils in asthma and other allergic diseases. Br. Med. Bull. 2000;56:985–1003. - PubMed
-
- Brightling CE, Bradding P, Symon FA, Holgate ST, Wardlaw AJ, Pavord ID. Mast-cell infiltration of airway smooth muscle in asthma. N. Engl. J. Med. 2002;346:1699–1705. - PubMed
-
- Brightling CE, Symon FA, Birring SS, Bradding P, Pavord ID, Wardlaw AJ. TH2 cytokine expression in bronchoalveolar lavage fluid T lymphocytes and bronchial submucosa is a feature of asthma and eosinophilic bronchitis. J. Allergy Clin. Immunol. 2002;110:899–905. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

