Transplanted astrocytes derived from BMP- or CNTF-treated glial-restricted precursors have opposite effects on recovery and allodynia after spinal cord injury
- PMID: 18803859
- PMCID: PMC2776404
- DOI: 10.1186/jbiol85
Transplanted astrocytes derived from BMP- or CNTF-treated glial-restricted precursors have opposite effects on recovery and allodynia after spinal cord injury
Abstract
Background: Two critical challenges in developing cell-transplantation therapies for injured or diseased tissues are to identify optimal cells and harmful side effects. This is of particular concern in the case of spinal cord injury, where recent studies have shown that transplanted neuroepithelial stem cells can generate pain syndromes.
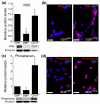
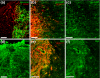
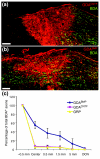
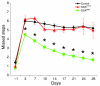
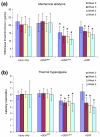
Results: We have previously shown that astrocytes derived from glial-restricted precursor cells (GRPs) treated with bone morphogenetic protein-4 (BMP-4) can promote robust axon regeneration and functional recovery when transplanted into rat spinal cord injuries. In contrast, we now show that transplantation of GRP-derived astrocytes (GDAs) generated by exposure to the gp130 agonist ciliary neurotrophic factor (GDAs(CNTF)), the other major signaling pathway involved in astrogenesis, results in failure of axon regeneration and functional recovery. Moreover, transplantation of GDA(CNTF) cells promoted the onset of mechanical allodynia and thermal hyperalgesia at 2 weeks after injury, an effect that persisted through 5 weeks post-injury. Delayed onset of similar neuropathic pain was also caused by transplantation of undifferentiated GRPs. In contrast, rats transplanted with GDAs(BMP) did not exhibit pain syndromes.
Conclusion: Our results show that not all astrocytes derived from embryonic precursors are equally beneficial for spinal cord repair and they provide the first identification of a differentiated neural cell type that can cause pain syndromes on transplantation into the damaged spinal cord, emphasizing the importance of evaluating the capacity of candidate cells to cause allodynia before initiating clinical trials. They also confirm the particular promise of GDAs treated with bone morphogenetic protein for spinal cord injury repair.
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

