Regulated catalysis of extracellular nucleotides by vascular CD39/ENTPD1 is required for liver regeneration
- PMID: 18804472
- PMCID: PMC2900919
- DOI: 10.1053/j.gastro.2008.07.025
Regulated catalysis of extracellular nucleotides by vascular CD39/ENTPD1 is required for liver regeneration
Abstract
Background & aims: Little is known about how endothelial cells respond to injury, regulate hepatocyte turnover and reconstitute the hepatic vasculature. We aimed to determine the effects of the vascular ectonucleotidase CD39 on sinusoidal endothelial cell responses following partial hepatectomy and to dissect purinergic and growth factor interactions in this model.
Methods: Parameters of liver injury and regeneration, as well as the kinetics of hepatocellular and sinusoidal endothelial cell proliferation, were assessed following partial hepatectomy in mice that do not express CD39, that do not express ATP/UTP receptor P2Y2, and in controls. The effects of extracellular ATP on vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), and interleukin-6 responses were determined in vivo and in vitro. Phosphorylation of the endothelial VEGF receptor in response to extracellular nucleotides and growth factors was assessed in vitro.
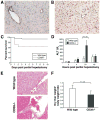
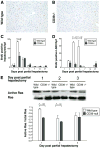
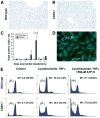
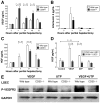
Results: After partial hepatectomy, expression of the vascular ectonucleotidase CD39 increased on sinusoidal endothelial cells. Targeted disruption of CD39 impaired hepatocellular regeneration, reduced angiogenesis, and increased hepatic injury, resulting in pronounced vascular endothelial apoptosis, and decreased survival. Decreased HGF release by sinusoidal endothelial cells, despite high levels of VEGF, reduced paracrine stimulation of hepatocytes. Failure of VEGF receptor-2/KDR transactivation by extracellular nucleotides on CD39-null endothelial cells was associated with P2Y2 receptor desensitization.
Conclusions: Regulated phosphohydrolysis of extracellular nucleotides by CD39 coordinates both hepatocyte and endothelial cell proliferation following partial hepatectomy. Lack of CD39 activity is associated with decreased hepatic regeneration and failure of vascular reconstitution.
Figures

Similar articles
-
CD39 modulates hematopoietic stem cell recruitment and promotes liver regeneration in mice and humans after partial hepatectomy.Ann Surg. 2013 Apr;257(4):693-701. doi: 10.1097/SLA.0b013e31826c3ec2. Ann Surg. 2013. PMID: 23474584 Free PMC article.
-
Inductive angiocrine signals from sinusoidal endothelium are required for liver regeneration.Nature. 2010 Nov 11;468(7321):310-5. doi: 10.1038/nature09493. Nature. 2010. PMID: 21068842 Free PMC article.
-
Deletion of cd39/entpd1 results in hepatic insulin resistance.Diabetes. 2008 Sep;57(9):2311-20. doi: 10.2337/db07-1265. Epub 2008 Jun 20. Diabetes. 2008. PMID: 18567823 Free PMC article.
-
The role of purinergic signaling in the liver and in transplantation: effects of extracellular nucleotides on hepatic graft vascular injury, rejection and metabolism.Front Biosci. 2008 Jan 1;13:2588-603. doi: 10.2741/2868. Front Biosci. 2008. PMID: 17981736 Free PMC article. Review.
-
The vascular endothelial growth factor signaling pathway regulates liver sinusoidal endothelial cells during liver regeneration after partial hepatectomy.Expert Rev Gastroenterol Hepatol. 2021 Feb;15(2):139-147. doi: 10.1080/17474124.2020.1815532. Epub 2020 Oct 14. Expert Rev Gastroenterol Hepatol. 2021. PMID: 32902336 Review.
Cited by
-
Synapomorphic features of hepatic and pulmonary vasculatures include comparable purinergic signaling responses in host defense and modulation of inflammation.Am J Physiol Gastrointest Liver Physiol. 2021 Aug 1;321(2):G200-G212. doi: 10.1152/ajpgi.00406.2020. Epub 2021 Jun 9. Am J Physiol Gastrointest Liver Physiol. 2021. PMID: 34105986 Free PMC article. Review.
-
P2Y2 nucleotide receptors mediate metalloprotease-dependent phosphorylation of epidermal growth factor receptor and ErbB3 in human salivary gland cells.J Biol Chem. 2010 Mar 5;285(10):7545-55. doi: 10.1074/jbc.M109.078170. Epub 2010 Jan 11. J Biol Chem. 2010. PMID: 20064929 Free PMC article.
-
Immune checkpoint modulators in cancer immunotherapy: recent advances and emerging concepts.J Hematol Oncol. 2022 Aug 17;15(1):111. doi: 10.1186/s13045-022-01325-0. J Hematol Oncol. 2022. PMID: 35978433 Free PMC article. Review.
-
Ion Channels and Oxidative Stress as a Potential Link for the Diagnosis or Treatment of Liver Diseases.Oxid Med Cell Longev. 2016;2016:3928714. doi: 10.1155/2016/3928714. Epub 2016 Jan 5. Oxid Med Cell Longev. 2016. PMID: 26881024 Free PMC article. Review.
-
Role of purinergic P2X receptors in the control of liver homeostasis.Wiley Interdiscip Rev Membr Transp Signal. 2012 May;1(3):341-348. doi: 10.1002/wmts.32. Epub 2012 Jan 11. Wiley Interdiscip Rev Membr Transp Signal. 2012. PMID: 22662313 Free PMC article.
References
-
- Fausto N, Campbell JS, Riehle KJ. Liver regeneration. Hepatology. 2006;43:S45–S53. - PubMed
-
- Michalopoulos GK, DeFrances MC. Liver regeneration. Science. 1997;276:60–66. - PubMed
-
- Taub R. Liver regeneration: from myth to mechanism. Nat Rev Mol Cell Biol. 2004;5:836–847. - PubMed
-
- LeCouter J, Moritz DR, Li B, et al. Angiogenesis-independent endothelial protection of liver: role of VEGFR-1. Science. 2003;299:890–893. - PubMed
-
- Lesurtel M, Graf R, Aleil B, et al. Platelet-derived serotonin mediates liver regeneration. Science. 2006;312:104–107. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

