Desensitization of the soluble guanylyl cyclase/cGMP pathway by lipopolysaccharide in rat isolated pulmonary artery but not aorta
- PMID: 18806822
- PMCID: PMC2607199
- DOI: 10.1038/bjp.2008.358
Desensitization of the soluble guanylyl cyclase/cGMP pathway by lipopolysaccharide in rat isolated pulmonary artery but not aorta
Abstract
Background and purpose: To investigate the function of soluble guanylyl cyclase (sGC)/3',5'-cyclic guanosine monophosphate (cGMP) pathway in lipopolysaccharide (LPS)-induced changes in vascular reactivity of rat isolated pulmonary artery and aorta.
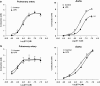
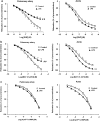
Experimental approach: Nitric oxide (NO) production, contraction responses to endothelin-1 (ET-1), relaxation responses to sodium nitroprusside (SNP), 8-pCPT-cGMP, BAY412272 and T-0156, SNP-induced cGMP production and expression of sGC(alpha1), sGC(beta1) and 3',5'-cyclic nucleotide phosphodiesterase-5 (PDE5) proteins were measured in LPS-treated pulmonary and aortic rings from male Wistar rats.
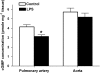
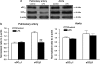
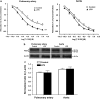
Key results: In both vessels, LPS (10 microg mL(-1), 20 h) increased NO production, which was inhibited by the selective inducible NOS (iNOS) inhibitor 1400W (1 microM). In the aorta, LPS decreased ET-1-induced contractility and this decrease was inhibited by the selective sGC inhibitor ODQ (10 microM) but not by removal of endothelium, or inhibitors of cyclooxygenase (indomethacin, 10 microM) or iNOS (1400W, 1 microM). Furthermore, aortic relaxation responses to the direct sGC activator BAY412272 were enhanced. In the pulmonary artery, SNP (1 nM to 30 microM)-induced relaxation and cGMP production, BAY412272-induced relaxation and sGC(beta1) protein expression were decreased, whereas relaxation responses to the PDE5-specific inhibitor T-0156 (0.1-100 nM) were enhanced. Relaxation responses to the phosphodiesterase-resistant cGMP analogue, 8-pCPT-cGMP, and protein expression levels of sGC(alpha1) and PDE5 were not altered in either vessel.
Conclusion and implications: LPS caused a selective hypocontractility of rat aorta to ET-1 mediated mainly through NO-independent sGC activation, whereas in the pulmonary artery, the effect of sGC activation was reduced by a decreased protein expression of sGC(beta1) together with increased PDE5 activity.
Figures

Similar articles
-
Escherichia coli lipopolysaccharide downregulates soluble guanylate cyclase in pulmonary artery smooth muscle.J Surg Res. 1998 Dec;80(2):309-14. doi: 10.1006/jsre.1998.5442. J Surg Res. 1998. PMID: 9878330
-
Nitric oxide-independent down-regulation of soluble guanylyl cyclase by bacterial endotoxin in astroglial cells.J Neurochem. 1999 Nov;73(5):2149-57. J Neurochem. 1999. PMID: 10537075
-
Effect of YC-1, an NO-independent, superoxide-sensitive stimulator of soluble guanylyl cyclase, on smooth muscle responsiveness to nitrovasodilators.Br J Pharmacol. 1997 Feb;120(4):681-9. doi: 10.1038/sj.bjp.0700982. Br J Pharmacol. 1997. PMID: 9051308 Free PMC article.
-
Nitric oxide-cyclic GMP pathway with some emphasis on cavernosal contractility.Int J Impot Res. 2004 Dec;16(6):459-69. doi: 10.1038/sj.ijir.3901256. Int J Impot Res. 2004. PMID: 15229623 Review.
-
The nitric oxide-soluble guanylate cyclase-cGMP pathway in pulmonary hypertension: from PDE5 to soluble guanylate cyclase.Eur Respir Rev. 2024 Mar 20;33(171):230183. doi: 10.1183/16000617.0183-2023. Print 2024 Jan 31. Eur Respir Rev. 2024. PMID: 38508664 Free PMC article. Review.
Cited by
-
Sodium nitroprusside restored lipopolysaccharide-induced learning and memory impairment in male rats via attenuating inflammation and oxidative stress.Physiol Rep. 2024 Jun;12(11):e16053. doi: 10.14814/phy2.16053. Physiol Rep. 2024. PMID: 38806440 Free PMC article.
-
Ropivacaine-induced contraction is attenuated by both endothelial nitric oxide and voltage-dependent potassium channels in isolated rat aortae.Biomed Res Int. 2013;2013:565271. doi: 10.1155/2013/565271. Epub 2013 Nov 20. Biomed Res Int. 2013. PMID: 24350275 Free PMC article.
-
Chronic nicotine treatment enhances vascular smooth muscle relaxation in rats.Acta Pharmacol Sin. 2015 Apr;36(4):429-39. doi: 10.1038/aps.2015.5. Epub 2015 Mar 23. Acta Pharmacol Sin. 2015. PMID: 25832423 Free PMC article.
References
-
- Bachetti T, Pasini E, Suzuki H, Ferrari R. Species-specific modulation of the nitric oxide pathway after acute experimentally induced endotoxemia. Crit Care Med. 2003;31:1509–1514. - PubMed
-
- Boerrigter G, Burnett JC., Jr Nitric oxide-independent stimulation of soluble guanylate cyclase with BAY 41-2272 in cardiovascular disease. Cardiovasc Drug Rev. 2007;25:30–45. - PubMed
-
- Chen SJ, Chen KH, Wu CC. Nitric oxide-cyclic GMP contributes to abnormal activation of Na+-K+-ATPase in the aorta from rats with endotoxic shock. Shock. 2005;23:179–185. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

