Late congestive heart failure after hematopoietic cell transplantation
- PMID: 18809605
- PMCID: PMC2651101
- DOI: 10.1200/JCO.2008.17.7428
Late congestive heart failure after hematopoietic cell transplantation
Abstract
Purpose: To examine the independent roles of pre-hematopoietic cell transplantation (HCT) therapeutic exposures, transplantation-related conditioning, and comorbidities (pre- and post-HCT) in the development of late congestive heart failure (CHF) after HCT.
Methods: This was a nested case-control design. Individuals with late CHF (diagnosed >or= 1 year after HCT) were identified from a cohort of 2,938 1+ year survivors who underwent transplantation at City of Hope National Medical Center, Duarte, CA. This cohort formed the sampling frame for selecting controls (without CHF) matched for age and year of HCT, donor source (allogeneic v autologous), and length of follow-up.
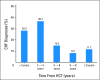
Results: Sixty patients with late CHF were identified; median age at HCT was 45.3 years (range, 16.6 to 68.6 years); median time to CHF was 3.0 years (range, 1.03 to 18.9 years); 68% received autologous HCT. Median ejection fraction was 36.9% (range, 15% to 53%). Compared with matched controls (n = 166), patients with late CHF received more cycles of pre-HCT chemotherapy (8.6 v 4.9 cycles; P < .01), had greater body mass index at HCT (28.4 v 26.2 kg/m(2); P = .01), greater lifetime anthracycline exposure (285.3 v 175.6 mg/m(2); P < .01), and were more likely to have multiple chronic comorbidities (30.0% v 13.9%; P < .01). Multivariable analysis revealed number of pre-HCT chemotherapy cycles (odds ratio [OR] = 1.2; P < .01), anthracycline dose >/= 250 mg/m(2) (OR = 3.2; P = .05), and two or more chronic comorbidities (OR = 4.3; P = .01) to be independently associated with late CHF.
Conclusion: Pre-HCT exposure to anthracyclines and presence of comorbidities are primarily responsible for the risk associated with late CHF after HCT. Conditioning-related therapeutic exposure does not contribute significantly to the risk. These results form the basis for identifying high-risk individuals for targeted surveillance, as well as developing preventive strategies in the form of aggressive management of comorbidities.
Figures
References
-
- Passweg JR, Rowlings PA, Armitage JO, et al: Report from the International Bone Marrow Transplant Registry and Autologous Blood and Marrow Transplant Registry–North America. Clin Transpl 117-127, 1995 - PubMed
-
- Horowitz MM, Rowlings PA: An update from the International Bone Marrow Transplant Registry and the Autologous Blood and Marrow Transplant Registry on current activity in hematopoietic stem cell transplantation. Curr Opin Hematol 4:395-400, 1997 - PubMed
-
- Wingard JR, Vogelsang GB, Deeg HJ: Stem cell transplantation: Supportive care and long-term complications. Hematology Am Soc Hematol Educ Program 422-444, 2002 - PubMed
-
- Socié G, Stone JV, Wingard JR, et al: Long-term survival and late deaths after allogeneic bone marrow transplantation: Late Effects Working Committee of the International Bone Marrow Transplant Registry. N Engl J Med 341:14-21, 1999 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical