Borrelia burgdorferi lacking DbpBA exhibits an early survival defect during experimental infection
- PMID: 18809667
- PMCID: PMC2583571
- DOI: 10.1128/IAI.00690-08
Borrelia burgdorferi lacking DbpBA exhibits an early survival defect during experimental infection
Abstract
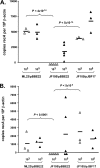
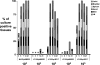
Several Borrelia burgdorferi genes induced under mammalian host conditions have been purported to be important in Lyme disease pathogenesis based on their binding to host structures. These genes include the dbpBA locus, whose products bind host decorin and glycosoaminoglycans. Recently, the dbpBA genes were reported to be involved in borrelial infectivity. Here we extended the previous observations by using culture and quantitative PCR to evaluate low- and high-dose murine infection by a Delta dbpBA::Gent(r) derivative of B. burgdorferi strain B31. The results indicate that the Delta dbpBA::Gent(r) mutant is attenuated in the ability to initially colonize and then persist in multiple tissues. The mutant exhibited a colonization defect as early as 3 days postinfection, before the development of an adaptive immune response, and after low-dose infection of SCID mice, which are deficient in adaptive immunity. These findings suggest that the inability to adhere to host decorin may promote clearance of B. burgdorferi, presumably via innate immune mechanisms. In a high-dose infection, the mutant disseminated to several tissues, particularly joint tissue, but it was generally cleared from these tissues by 3 weeks postinfection. Finally, following high-dose infection of SCID mice, the dbpBA mutant exhibited only a mild colonization defect, suggesting that the adaptive response is involved in the clearance of the mutant in immunocompetent mice. Taken together, these results suggest that the DbpBA proteins facilitate the colonization of multiple tissues by B. burgdorferi and are required for optimal resistance to both innate and adaptive immune mechanisms following needle inoculation.
Figures


Similar articles
-
Assessment of decorin-binding protein A to the infectivity of Borrelia burgdorferi in the murine models of needle and tick infection.BMC Microbiol. 2008 May 28;8:82. doi: 10.1186/1471-2180-8-82. BMC Microbiol. 2008. PMID: 18507835 Free PMC article.
-
The dbpBA locus of Borrelia burgdorferi is not essential for infection of mice.Infect Immun. 2006 Nov;74(11):6509-12. doi: 10.1128/IAI.00740-06. Epub 2006 Sep 5. Infect Immun. 2006. PMID: 16954404 Free PMC article.
-
Both decorin-binding proteins A and B are critical for the overall virulence of Borrelia burgdorferi.Infect Immun. 2008 Mar;76(3):1239-46. doi: 10.1128/IAI.00897-07. Epub 2008 Jan 14. Infect Immun. 2008. PMID: 18195034 Free PMC article.
-
Adhesion mechanisms of Borrelia burgdorferi.Adv Exp Med Biol. 2011;715:35-49. doi: 10.1007/978-94-007-0940-9_3. Adv Exp Med Biol. 2011. PMID: 21557056 Free PMC article. Review.
-
Borrelia burgdorferi Pathogenesis and the Immune Response.Clin Lab Med. 2015 Dec;35(4):745-64. doi: 10.1016/j.cll.2015.07.004. Epub 2015 Aug 8. Clin Lab Med. 2015. PMID: 26593255 Review.
Cited by
-
Illuminating the roles of the Borrelia burgdorferi adhesins.Trends Microbiol. 2013 Aug;21(8):372-9. doi: 10.1016/j.tim.2013.06.005. Epub 2013 Jul 19. Trends Microbiol. 2013. PMID: 23876218 Free PMC article. Review.
-
The role of Borrelia burgdorferi outer surface proteins.FEMS Immunol Med Microbiol. 2012 Oct;66(1):1-19. doi: 10.1111/j.1574-695X.2012.00980.x. Epub 2012 May 21. FEMS Immunol Med Microbiol. 2012. PMID: 22540535 Free PMC article. Review.
-
Cyclic di-GMP modulates gene expression in Lyme disease spirochetes at the tick-mammal interface to promote spirochete survival during the blood meal and tick-to-mammal transmission.Infect Immun. 2015 Aug;83(8):3043-60. doi: 10.1128/IAI.00315-15. Epub 2015 May 18. Infect Immun. 2015. PMID: 25987708 Free PMC article.
-
In Vivo Imaging Demonstrates That Borrelia burgdorferi ospC Is Uniquely Expressed Temporally and Spatially throughout Experimental Infection.PLoS One. 2016 Sep 9;11(9):e0162501. doi: 10.1371/journal.pone.0162501. eCollection 2016. PLoS One. 2016. PMID: 27611840 Free PMC article.
-
Decorin binding proteins of Borrelia burgdorferi promote arthritis development and joint specific post-treatment DNA persistence in mice.PLoS One. 2015 Mar 27;10(3):e0121512. doi: 10.1371/journal.pone.0121512. eCollection 2015. PLoS One. 2015. PMID: 25816291 Free PMC article.
References
-
- Akins, D. R., S. F. Porcella, T. G. Popova, D. Shevchenko, S. I. Baker, M. Li, M. V. Norgard, and J. D. Radolf. 1995. Evidence for in vivo but not in vitro expression of a Borrelia burgdorferi outer surface protein F (OspF) homologue. Mol. Microbiol. 18507-520. - PubMed
-
- Anguita, J., N. Ramamoorthi, J. W. Hovius, S. Das, V. Thomas, R. Persinski, D. Conze, P. W. Askenase, M. Rincon, F. S. Kantor, and E. Fikrig. 2002. Salp15, an Ixodes scapularis salivary protein, inhibits CD4+ T cell activation. Immunity 16849-859. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

