NCAM induces CaMKIIalpha-mediated RPTPalpha phosphorylation to enhance its catalytic activity and neurite outgrowth
- PMID: 18809727
- PMCID: PMC2542478
- DOI: 10.1083/jcb.200803045
NCAM induces CaMKIIalpha-mediated RPTPalpha phosphorylation to enhance its catalytic activity and neurite outgrowth
Abstract
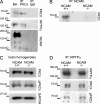
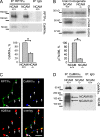
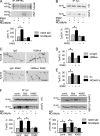
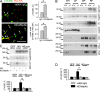
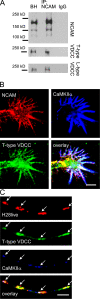
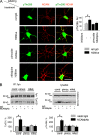
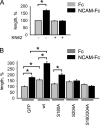
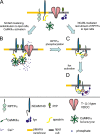
Receptor protein tyrosine phosphatase alpha (RPTPalpha) phosphatase activity is required for intracellular signaling cascades that are activated in motile cells and growing neurites. Little is known, however, about mechanisms that coordinate RPTPalpha activity with cell behavior. We show that clustering of neural cell adhesion molecule (NCAM) at the cell surface is coupled to an increase in serine phosphorylation and phosphatase activity of RPTPalpha. NCAM associates with T- and L-type voltage-dependent Ca(2+) channels, and NCAM clustering at the cell surface results in Ca(2+) influx via these channels and activation of NCAM-associated calmodulin-dependent protein kinase IIalpha (CaMKIIalpha). Clustering of NCAM promotes its redistribution to lipid rafts and the formation of a NCAM-RPTPalpha-CaMKIIalpha complex, resulting in serine phosphorylation of RPTPalpha by CaMKIIalpha. Overexpression of RPTPalpha with mutated Ser180 and Ser204 interferes with NCAM-induced neurite outgrowth, which indicates that neurite extension depends on NCAM-induced up-regulation of RPTPalpha activity. Thus, we reveal a novel function for a cell adhesion molecule in coordination of cell behavior with intracellular phosphatase activity.
Figures


References
-
- Beggs, H.E., S.C. Baragona, J.J. Hemperly, and P.F. Maness. 1997. NCAM140 interacts with the focal adhesion kinase p125(fak) and the SRC-related tyrosine kinase p59(fyn). J. Biol. Chem. 272:8310–8319. - PubMed
-
- Bhandari, V., K.L. Lim, and C.J. Pallen. 1998. Physical and functional interactions between receptor-like protein-tyrosine phosphatase alpha and p59fyn. J. Biol. Chem. 273:8691–8698. - PubMed
-
- Blanchetot, C., and J. den Hertog. 2000. Multiple interactions between receptor protein-tyrosine phosphatase (RPTP) alpha and membrane-distal protein-tyrosine phosphatase domains of various RPTPs. J. Biol. Chem. 275:12446–12452. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

