Loss of p120 catenin and links to mitotic alterations, inflammation, and skin cancer
- PMID: 18809907
- PMCID: PMC2547465
- DOI: 10.1073/pnas.0807301105
Loss of p120 catenin and links to mitotic alterations, inflammation, and skin cancer
Abstract
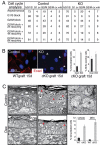
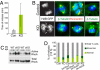
Tumor formation involves epigenetic modifications and microenvironmental changes as well as cumulative genetic alterations encompassing somatic mutations, loss of heterozygosity, and aneuploidy. Here, we show that conditional targeting of p120 catenin in mice leads to progressive development of skin neoplasias associated with intrinsic NF-kappaB activation. We find that, similarly, squamous cell carcinomas in humans display altered p120 and activated NF-kappaB. We show that epidermal hyperproliferation arising from p120 loss can be abrogated by IkappaB kinase 2 inhibitors. Although this underscores the importance of this pathway, the role of NF-kappaB in hyperproliferation appears rooted in its impact on epidermal microenvironment because as p120-null keratinocytes display a growth-arrested phenotype in culture. We trace this to a mitotic defect, resulting in unstable, binucleated cells in vitro and in vivo. We show that the abnormal mitoses can be ameliorated by inhibiting RhoA, the activity of which is abnormally high. Conversely, we can elicit such mitotic defects in control keratinocytes by elevating RhoA activity. The ability of p120 deficiency to elicit mitotic alterations and chronic inflammatory responses, that together may facilitate the development of genetic instability in vivo, provides insights into why it figures so prominently in skin cancer progression.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Comment in
-
Dissecting the role of cadherin-catenin proteins in mammalian epidermis.Proc Natl Acad Sci U S A. 2008 Oct 7;105(40):15225-6. doi: 10.1073/pnas.0808458105. Epub 2008 Oct 1. Proc Natl Acad Sci U S A. 2008. PMID: 18832145 Free PMC article. No abstract available.
References
-
- Birchmeier W, Behrens J. Cadherin expression in carcinomas: Role in the formation of cell junctions and the prevention of invasiveness. Biochim Biophys Acta. 1994;1198:11–26. - PubMed
-
- Reynolds AB, Roczniak-Ferguson A. Emerging roles for p120-catenin in cell adhesion and cancer. Oncogene. 2004;23:7947–7956. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

