Calcium orthophosphates: crystallization and dissolution
- PMID: 18816145
- PMCID: PMC2743557
- DOI: 10.1021/cr0782574
Calcium orthophosphates: crystallization and dissolution
Figures










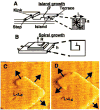
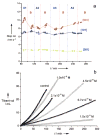




References
-
- Dorozhkin SV. J Mater Sci. 2007;42:1061. - PubMed
-
- Koutsoukos PG, Amjad Z, Tomson MB, Nancollas GH. J Am Chem Soc. 1980;102:1553.
-
- Johnsson MSA, Nancollas GH. Crit Rev Oral Biol Med. 1992;3:61. - PubMed
-
- Dorozhkin SV, Epple M. Angew Chem, Int Ed. 2002;41:3130. - PubMed
-
- Elliot JC. Structure and Chemistry of the Apatites and Other Calcium Orthosphosphates. Elsevier: Amsterdam; 1994.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

