Population pharmacokinetic and pharmacogenetic analysis of 6-mercaptopurine in paediatric patients with acute lymphoblastic leukaemia
- PMID: 18823306
- PMCID: PMC2675766
- DOI: 10.1111/j.1365-2125.2008.03281.x
Population pharmacokinetic and pharmacogenetic analysis of 6-mercaptopurine in paediatric patients with acute lymphoblastic leukaemia
Abstract
Aims: To investigate the population pharmacokinetics of 6-mercaptopurine (6-MP) active metabolites in paediatric patients with acute lymphoblastic leukaemia (ALL) and examine the effects of various genetic polymorphisms on the disposition of these metabolites.
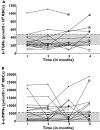
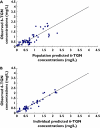
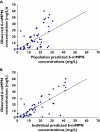
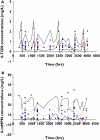
Methods: Data were collected prospectively from 19 paediatric patients with ALL (n = 75 samples, 150 concentrations) who received 6-MP maintenance chemotherapy (titrated to a target dose of 75 mg m(-2) day(-1)). All patients were genotyped for polymorphisms in three enzymes involved in 6-MP metabolism. Population pharmacokinetic analysis was performed with the nonlinear mixed effects modelling program (nonmem) to determine the population mean parameter estimate of clearance for the active metabolites.
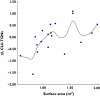
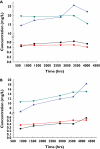
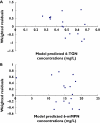
Results: The developed model revealed considerable interindividual variability (IIV) in the clearance of 6-MP active metabolites [6-thioguanine nucleotides (6-TGNs) and 6-methylmercaptopurine nucleotides (6-mMPNs)]. Body surface area explained a significant part of 6-TGNs clearance IIV when incorporated in the model (IIV reduced from 69.9 to 29.3%). The most influential covariate examined, however, was thiopurine methyltransferase (TPMT) genotype, which resulted in the greatest reduction in the model's objective function (P < 0.005) when incorporated as a covariate affecting the fractional metabolic transformation of 6-MP into 6-TGNs. The other genetic covariates tested were not statistically significant and therefore were not included in the final model.
Conclusions: The developed pharmacokinetic model (if successful at external validation) would offer a more rational dosing approach for 6-MP than the traditional empirical method since it combines the current practice of using body surface area in 6-MP dosing with a pharmacogenetically guided dosing based on TPMT genotype.
Figures
Similar articles
-
A comparison of red blood cell thiopurine metabolites in children with acute lymphoblastic leukemia who received oral mercaptopurine twice daily or once daily: a Pediatric Oncology Group study (now The Children's Oncology Group).Pediatr Blood Cancer. 2004 Aug;43(2):105-9. doi: 10.1002/pbc.20089. Pediatr Blood Cancer. 2004. PMID: 15236274 Clinical Trial.
-
Thiopurine S-methyltransferase (TPMT) polymorphisms in children with acute lymphoblastic leukemia, and the need for reduction or cessation of 6-mercaptopurine doses during maintenance therapy: the Polish multicenter analysis.Pediatr Blood Cancer. 2011 Oct;57(4):578-82. doi: 10.1002/pbc.23013. Epub 2011 Feb 11. Pediatr Blood Cancer. 2011. PMID: 21319286
-
Thiopurine S-methyltransferase (TPMT) gene polymorphism in Brazilian children with acute lymphoblastic leukemia: association with clinical and laboratory data.Ther Drug Monit. 2008 Dec;30(6):700-4. doi: 10.1097/FTD.0b013e31818b0f31. Ther Drug Monit. 2008. PMID: 19057372
-
Individualization of thiopurine therapy: thiopurine S-methyltransferase and beyond.Pharmacogenomics. 2009 Aug;10(8):1309-22. doi: 10.2217/pgs.09.78. Pharmacogenomics. 2009. PMID: 19663675 Review.
-
[New techniques for optimization of thiopurine therapy in leukemia and transplantation].Tidsskr Nor Laegeforen. 2002 Apr 30;122(11):1107-10. Tidsskr Nor Laegeforen. 2002. PMID: 12043054 Review. Norwegian.
Cited by
-
Genotypes Affecting the Pharmacokinetics of Anticancer Drugs.Clin Pharmacokinet. 2017 Apr;56(4):317-337. doi: 10.1007/s40262-016-0450-z. Clin Pharmacokinet. 2017. PMID: 27641154 Free PMC article. Review.
-
Prediction of leukocyte counts during paediatric acute lymphoblastic leukaemia maintenance therapy.Sci Rep. 2019 Dec 2;9(1):18076. doi: 10.1038/s41598-019-54492-5. Sci Rep. 2019. PMID: 31792398 Free PMC article.
-
The development of an objective methodology to measure medication adherence to oral thiopurines in paediatric patients with acute lymphoblastic leukaemia--an exploratory study.Eur J Clin Pharmacol. 2009 Nov;65(11):1105-12. doi: 10.1007/s00228-009-0700-1. Epub 2009 Jul 28. Eur J Clin Pharmacol. 2009. PMID: 19636546
-
Impact of the loss of slc43a3 on 6-mercaptopurine absorption and tissue distribution in mice.Drug Metab Dispos. 2025 Apr;53(4):100054. doi: 10.1016/j.dmd.2025.100054. Epub 2025 Mar 3. Drug Metab Dispos. 2025. PMID: 40133022 Free PMC article.
-
Model-Based Individualized Treatment of Chemotherapeutics: Bayesian Population Modeling and Dose Optimization.PLoS One. 2015 Jul 30;10(7):e0133244. doi: 10.1371/journal.pone.0133244. eCollection 2015. PLoS One. 2015. PMID: 26226448 Free PMC article.
References
-
- Lennard L, Keen D, Lilleyman JS. Oral 6-mercaptopurine in childhood leukemia: parent drug pharmacokinetics and active metabolite concentrations. Clin Pharmacol Ther. 1986;40:287–92. - PubMed
-
- Lennard L. The clinical pharmacology of 6-mercaptopurine. Eur J Clin Pharmacol. 1992;43:329–39. - PubMed
-
- Derijks LJ, Gilissen LP, Hooymans PM, Hommes DW. Review article: thiopurines in inflammatory bowel disease. Aliment Pharmacol Ther. 2006;24:715–29. - PubMed
-
- Fairchild CR, Maybaum J, Kennedy KA. Concurrent unilateral chromatid damage and DNA strand breakage in response to 6-thioguanine treatment. Biochem Pharmacol. 1986;35:3533–41. - PubMed
-
- Elion GB. The purine path to chemotherapy. Science. 1989;244:41–7. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

