COX-2 expression and function in the hyperalgesic response to paw inflammation in mice
- PMID: 18829279
- PMCID: PMC2615797
- DOI: 10.1016/j.plefa.2008.08.001
COX-2 expression and function in the hyperalgesic response to paw inflammation in mice
Abstract
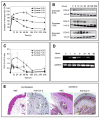
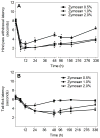
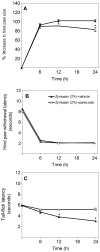
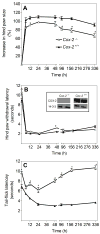
Peripheral inflammation and edema are often accompanied by primary and secondary hyperalgesia which are mediated by both peripheral and central mechanisms. The role of cyclooxygenase-2 (COX-2)-mediated prostanoid production in hyperalgesia is a topic of substantial current interest. We have established a murine foot-pad inflammation model in which both pharmacologic and genetic tools can be used to characterize the role of COX-2 in hyperalgesia. Zymosan, an extract from yeast, injected into the plantar surface of the hindpaw induces an edema response and an increase in COX-2 expression in the hindpaw, spinal cord and brain. Zymosan-induced primary hyperalgesia, measured as a decrease in hindpaw withdrawal latency in response to a thermal stimulus, is long-lasting and is not inhibited by pre-treatment with the systemic COX-2 selective inhibitor, parecoxib (20 mg/kg). In contrast, the central component of hyperalgesia, measured as a reduction in tail flick latency in response to heat, is reduced by parecoxib. Zymosan-induced primary hyperalgesia in Cox-2-/- mice is similar to that of their Cox-2+/+ littermate controls. However, the central component of hyperalgesia is substantially reduced in Cox-2-/- versus Cox-2+/+ mice, and returns to baseline values much more rapidly. Thus pharmacological data suggest, and genetic experiments confirm, (i) that primary hyperalgesia in response to zymosan inflammation in the mouse paw is not mediated by COX-2 function and (ii) that COX-2 function plays a major role in the central component of hyperalgesia in this model of inflammation.
Figures

Similar articles
-
Selective inhibitors of cyclo-oxygenase-2 (COX-2) induce hypoalgesia in a rat paw model of inflammation.Br J Pharmacol. 2002 Nov;137(6):837-44. doi: 10.1038/sj.bjp.0704937. Br J Pharmacol. 2002. PMID: 12411415 Free PMC article.
-
Sinularin from indigenous soft coral attenuates nociceptive responses and spinal neuroinflammation in carrageenan-induced inflammatory rat model.Mar Drugs. 2012 Sep;10(9):1899-1919. doi: 10.3390/md10091899. Epub 2012 Aug 24. Mar Drugs. 2012. PMID: 23118711 Free PMC article.
-
Suppressed injury-induced rise in spinal prostaglandin E2 production and reduced early thermal hyperalgesia in iNOS-deficient mice.J Neurosci. 2000 Sep 1;20(17):6714-20. doi: 10.1523/JNEUROSCI.20-17-06714.2000. J Neurosci. 2000. PMID: 10964977 Free PMC article.
-
Role of interleukin-1beta and tumor necrosis factor-alpha-dependent expression of cyclooxygenase-2 mRNA in thermal hyperalgesia induced by chronic inflammation in mice.Neuroscience. 2008 Mar 18;152(2):477-86. doi: 10.1016/j.neuroscience.2007.10.039. Epub 2007 Nov 12. Neuroscience. 2008. PMID: 18262365
-
[Analgesic effect of calpain inhibitor ALLN on the zymosan-induced paw inflammatory pain and its effect on the expression of cyclooxygenase-2 in the spinal dorsal horn].Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2012 Feb;34(1):25-31. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2012. PMID: 22737715 Chinese.
Cited by
-
Feedback regulation of cyclooxygenase-2 transcription ex vivo and in vivo.Biochem Biophys Res Commun. 2009 Jan 16;378(3):534-8. doi: 10.1016/j.bbrc.2008.11.099. Epub 2008 Dec 4. Biochem Biophys Res Commun. 2009. PMID: 19061862 Free PMC article.
-
Progress in PET Imaging of Neuroinflammation Targeting COX-2 Enzyme.Molecules. 2021 May 27;26(11):3208. doi: 10.3390/molecules26113208. Molecules. 2021. PMID: 34071951 Free PMC article. Review.
-
Nanoliposome-Encapsulated and Non-Encapsulated Phenolics From Achillea millefolium and Their Biological Function in Mice Challenged by Campylobacter jejuni: A Comparative Study.Front Mol Biosci. 2022 Feb 2;8:832022. doi: 10.3389/fmolb.2021.832022. eCollection 2021. Front Mol Biosci. 2022. PMID: 35187077 Free PMC article.
-
Long-Lasting Anti-Inflammatory and Antinociceptive Effects of Acute Ammonium Glycyrrhizinate Administration: Pharmacological, Biochemical, and Docking Studies.Molecules. 2019 Jul 4;24(13):2453. doi: 10.3390/molecules24132453. Molecules. 2019. PMID: 31277398 Free PMC article.
-
Anti-Inflammatory and Antinociceptive Properties of the Quercetin-3-Oleate AV2, a Novel FFAR1 Partial Agonist.Int J Mol Sci. 2024 Oct 30;25(21):11635. doi: 10.3390/ijms252111635. Int J Mol Sci. 2024. PMID: 39519187 Free PMC article.
References
-
- Smith WL, DeWitt DL, Garavito RM. Cyclooxygenases: structural, cellular and molecular biology. Ann Rev Biochem. 2000;69:145–182. - PubMed
-
- Herschman HR, Talley JJ, DuBois R. Cyclooxygenase 2 (COX-2) as a target for therapy and noninvasive imaging. Mol Imag Biol. 2003;5:286–303. - PubMed
-
- Ebersberger A, Grubb BD, Willingale HL, Gardiner NJ, Nebe J, Schaible HG. The intraspinal release of prostaglandin E2 in a model of acute arthritis is accompanied by an upregulation of cyclooxygenase-2 in the spinal cord. Neuroscience. 1999;93:775–781. - PubMed
-
- Ibuki T, Matsumura K, Yamazaki Y, Nozaki T, Tanaka Y, Kobayashi S. Cyclooxygenase-2 is induced in the endothelial cells throughout the central nervous system during carrageenan-induced hind paw inflammation; its possible role in hyperalgesia. J Neurochem. 2003;86:318–328. - PubMed
-
- Guay J, Bateman K, Gordon R, Mancini J, Riendeau D. Carrageenan-induced paw edema in rat elicits a predominant prostaglandin E2 (PGE2) response in the central nervous system associated with the induction of microsomal PGE2 synthase-1. J Biol Chem. 2004;279:24866–24872. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

